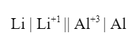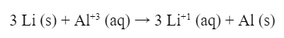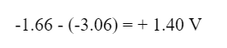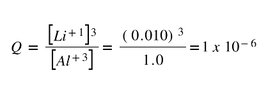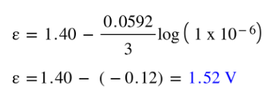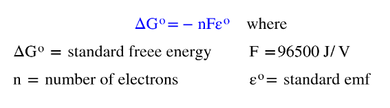Non Standard Voltage
|
Not all batteries are standard. In fact, most aren't. Standard means that the battery operation is happening at 298 K and that all solutions within the voltaic cells are 1.0 M. However, you can manipulate a battery's strength by changing the concentrations of the solutions.
Suppose you wanted to make an lithium/aluminum battery. First, the battery would have the following notation: Which means that the equation for the battery would be
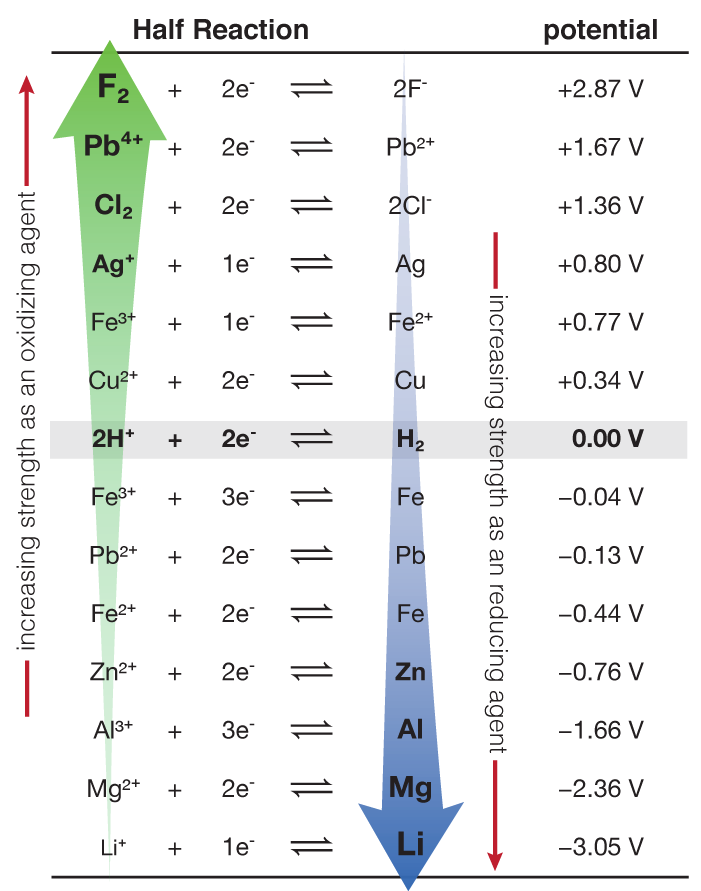
And the standard voltage would be
But what if it were not standard? Again, standard conditions for electrochemistry is defined as 298 K and 1.0 M solutions of all solutions. What if we changed the concentration of the solutions? That would affect the voltage of the battery if there was not enough or too many ions in the solutions of the two voltaic cells.
|
Through manipulations of the standard versus non standard free energy equations (sections 17.7 & 17.8), we can arrive at the equation:
Example: Let's say we make a lithium/aluminum battery, but instead of using standard 1 M solutions, we use a 1.0 M solution of Al(NO3)3 (aq) and a 0.010 M solution of LiNO3. What would be the voltage of this nonstandard battery?
|
To determine this we need to determine the n and Q.
n is the number of electrons transferred. Since aluminum goes from Al+3 (aq) to neutral Al, there is a transfer of 3 electrons.
And Q is reaction quotient (section 13.5), so products over reactants for all the aqueous solutions. Solids are left out because there concentrations do not change.
|
Free Energy and Voltage
Because some batteries work really well and others don't, there is actually a relationship between the voltage of a battery and its spontaneity. The higher the voltage, the more the electrons want to move from one electrode to the other, and the higher the spontaneity. Through manipulations of the work formula, the free energy equation and the energy/volt relationship, it was determined the relationship between free energy and voltage to be :
In the lithium/aluminum battery we have been using today, we can determine the standard free energy of the battery using information we have already gleaned earlier. Since the number of electrons transferred is still 3, F is 96,500 J/V (Faraday's constant) and the voltage is 1.40 V, then:
The fact that the standard free energy is a negative value reinforces that this is an effective battery. As we know, a positive voltage means that the battery will conduct a current through the cell. When the cell voltage is zero, that means the free energy will also be zero, making for a "dead" battery. When the cell voltage is negative, we are said to be "recharging" the battery. This is a nonspontaneous reaction, which will have a positive free energy.
Start @ 4:13
Start @ 4:13