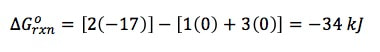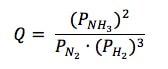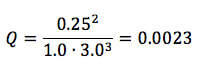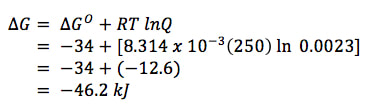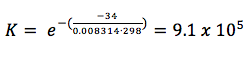Free Energy Applications
Non Standard Free Energy
Standard free energy, ΔGO, is determined when the temperature is at 298 K and all the products and reactants have a concentration of 1. Non standard free energy, ΔG, is when either the temperature is not 298 K, the concentrations are not 1, or both. Ordinarily, we do not perform experiments at standard conditions, so having an adaptation to the calculation for ΔG is helpful. When conditions are not standard, we can calculate the non standard free energy using this equation:
ΔG = ΔGO + RT lnQ
where RT lnQ is a fudge factor to adjust the standard free energy to non standard conditions. R is the gas law constant, but you must use 8.314 J/mol K, and you must have it in kJ. So, really, you need to use 8.314 x 10-3 kJ/mol K. T is the Kelvin temperature and Q is the reaction quotient, based on the equilibrium constant.
Example: Calculate ΔG for the reaction
Example: Calculate ΔG for the reaction
N2 (g) + 3 H2 (g) --> 2 NH3 (g)
at 250 K when 1.0 atm of N2, 3.0 atm of H2 and 0.25 atm of NH3 are placed in a container to react.
|
To find ΔGO, we use the standard numbers from the back of the book, and subtract the products from the reactants:
ΔGONH3 = -17 kJ/mol ΔGON2 = 0 kJ/mol ΔGOH2 = 0 kJ/mol Then, to find Q, it is like finding the equilibrium constant (Kp), where we take products over reactants to get the equation:
|
So, we have determined all the factors in the equation:
ΔG = ? ΔGO = -34 kJ R = 8.314 x 10-3 kJ/mol K T = 250 K Q = 0.0023 and when we insert numbers, we get
|
Relationship between Free Energy and Equilibrium
As we saw in the above equation, there is a relationship between free energy and equilibrium. We already know that when ΔG is 0, the system is at equilibrium. We can use this information to resolve the equation above for K, the equilibrium constant:
ΔGO = -RT ln K
or
K = e-ΔGO/RT
or
K = e-ΔGO/RT
Example 2: In example 1, we determined the standard free energy of the synthesis of NH3. What is the equilibrium constant for the reaction at standard conditions?