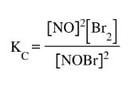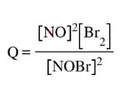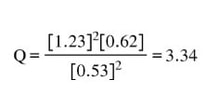Applications of Equilibrium
Reaction Quotient
Reaction quotient is a method to determine whether a chemical reaction is at equilibrium. Often, it is quite difficult to determine if a chemical reaction has achieved equilibrium. Especially if we are dealing with colorless gases or solutions. Meaning, we are not sure if the chemical reaction is at equilibrium or not. What we can do is measure the concentrations or partial pressures of all the materials in the reaction, and calculate the equilibrium constant as if the reaction was at equilibrium. When we calculate an equibrium constant and we are unsure as to whether the reaction is at equilibrium or not, we call it a reaction quotient and use the symbol Q instead of Kc or Kp.
Reaction quotient is a method to determine whether a chemical reaction is at equilibrium. Often, it is quite difficult to determine if a chemical reaction has achieved equilibrium. Especially if we are dealing with colorless gases or solutions. Meaning, we are not sure if the chemical reaction is at equilibrium or not. What we can do is measure the concentrations or partial pressures of all the materials in the reaction, and calculate the equilibrium constant as if the reaction was at equilibrium. When we calculate an equibrium constant and we are unsure as to whether the reaction is at equilibrium or not, we call it a reaction quotient and use the symbol Q instead of Kc or Kp.
This is easier to show by using an example. Let's say we are doing the following chemical reaction:
2 NOBr (g) <==> 2 NO (g) + Br2 (g) KC = 0.50
We are doing this experiment and the reaction is sitting there, and we don't know if it is at equilibrium or not. So, we measure the concentration of each of the gases. We determine the concentrations of each of the gases to be:
[NOBr] = 0.53 M
[NO] = 1.23 M
[Br2] = 0.62 M
If we were to calculate the Kc for this reaction we would need the following expression:
But, in this case, we don't know if the numbers we have are at equilibrium or not, so we replace the Kc with Q (reaction quotient):
We will input the numbers collected from the flask into the reaction quotient expression and calculate the Q.
In this case, the Q = 3.34. The Kc for this reaction is given to be 0.50. Since these are not the same, we know the system is not at equilibrium. But, which way is it going? Obviously, that question is rhetorical. I'm going to answer for you. Now, we notice that the the Q is actually 3.34, which is way bigger than 0.50. Since the number is too big to be at equilibrium, the chemical reaction must have too much of what is on the top of the fraction in the Q/Kc calculation and not enough of the bottom. That means there are too many products, and not enough reactants. Since this is the case, the chemical reaction needs to shift left, toward the reactants, to make more reactants from the products to get to equilibrium. In this case, the reaction is not at equilibrium and needs to shift to the reactants to achieve equilibrium.
In other words, by determining what the ratio of concentrations of the products and reactants are at any time, we can compare that value (Q) to the actual equilibrium constant (Kc or Kp), and determine whether the reaction is at equilibrium or not. And if the reaction is not at equilibrium, we should be able to tell which way it should go by these relationships:
Q = Kc, system is at equilibrium
Q > Kc, too many products, shift to the left
Q<Kc, too many reactants, shift to the right
Q > Kc, too many products, shift to the left
Q<Kc, too many reactants, shift to the right