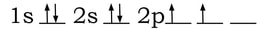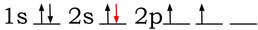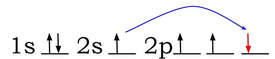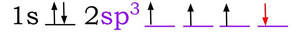Hybrid Orbitals
Limitations of VSEPR Theory
The VSEPR theory for how elements bond with one another does not fit with what we know about orbitals. In other words, the shapes that molecules form are not the same as an s or p orbital shape. So, how do orbitals and bonding shape coexist?
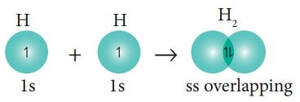
So, first of all, we think that bonding occurs when orbitals overlap. Meaning, in order for compounds to bond, the orbital of one atom has to overlap with the orbital of the other atom, so that the electrons shared are within the orbitals of both elements. For instance, in a molecule of hydrogen, it would look something like this:
The VSEPR theory for how elements bond with one another does not fit with what we know about orbitals. In other words, the shapes that molecules form are not the same as an s or p orbital shape. So, how do orbitals and bonding shape coexist?
So, first of all, we think that bonding occurs when orbitals overlap. Meaning, in order for compounds to bond, the orbital of one atom has to overlap with the orbital of the other atom, so that the electrons shared are within the orbitals of both elements. For instance, in a molecule of hydrogen, it would look something like this:
|
In this molecule, when the two 1s hydrogens come together, the 1s orbitals overlap, and the electrons "stay" in that overlap area to create the bond. This molecule would be linear, which is OK with our knowledge of atom orbitals.
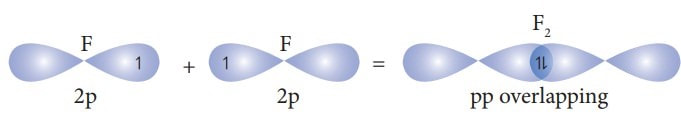
Similarly, in a diatomic molecule of fluorine, the lone 2p orbital of one fluorine can overlap with the lone 2p orbital of the other fluorine to create a bond. |
Hybrid Orbitals
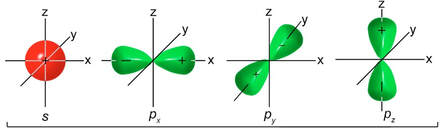
To explain more complicated molecule shapes, like tetrahedral, we use the idea of hybrid orbitals. Hybrid orbitals are the result of s and p (and sometimes d) orbitals combining properties within an atom to form new orbitals that are different shapes from the original shape of the s and p orbitals. For instance, carbon in a compound usually forms a tetrahedral shape with the elements it bonds with. Carbon has an electron configuration of 1s22s22p2. The orbital diagram would look like this:
When carbon bonds, the 2s and the 2 p orbitals perform a change that hybridizes them. One of the electrons from the 2s orbital (colored in red is promoted:
And then the s and p hybridize (combine and adjust their shape) to sp3 hybrid orbital:
Pictorally, it would start this way:
And, the hybridization would become:
|
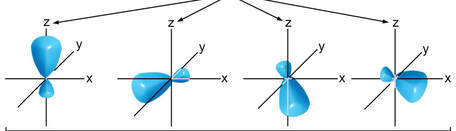
And when you combine the 4 hybrid orbitals into one shape, you get the tetrahedral shape you see at the right. Now, elements can come in and overlap their orbitals with the sp3 hybrid orbital to create a tetrahedral shape.
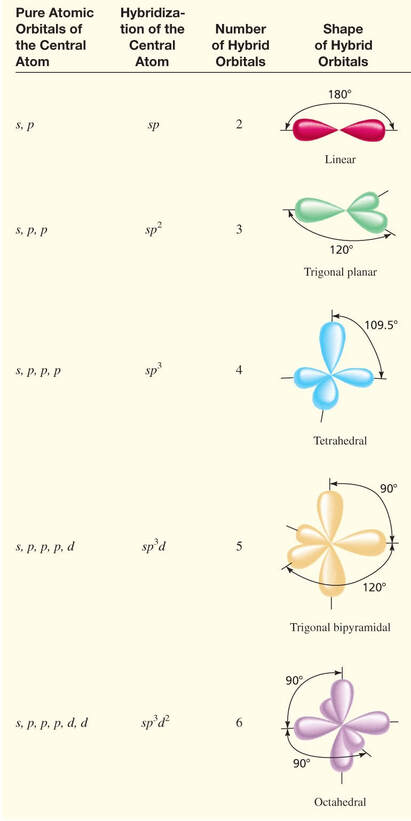
There are other hybrid orbitals, that are responsible for the shapes we witness. There is sp (linear), sp2 (trigonal planar), sp3 (tetrahedral), sp3d (trigonal bipyramidal) and sp3d2 (octahedral). Below is a picture of each of the hybrid orbitals. |
Multiple Bonds
So, how do multiple bonds work? How does that overlapping of orbitals work. Here's what we think.
So, how do multiple bonds work? How does that overlapping of orbitals work. Here's what we think.
|
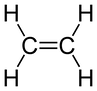
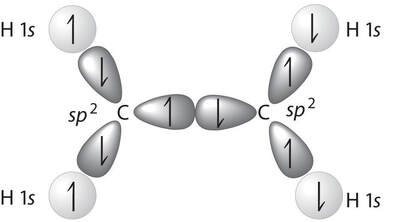
In C2H4, there is a double bond between the carbons. In the case of a double bond, we believe that the hybridization that occurs happens but, one less time. For instance, instead of carbon hybridizing sp3, it hybridizes sp2, leaving one p orbital subshell unhybridized:
This first overlap is called a sigma (σ) bond. |
|
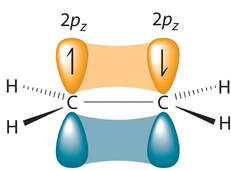
Then, the up and down p orbital, shown here in orange and blue, are drawn in close enough by the first bond to overlap. This is called a pi (π) bond. It looks like there are two overlaps here (one for the orange, one for the blue), but it's really only one bond, because there is only one electron in the orbital.
To simplify it as easy as possible: The first (or a single) bond between two atoms is called a σ bond. Any additional bond after that is called a π bond. In ethene, there are 5 σ bonds (four C-H bonds and the first C-C bond) and 1 π bond (the second C-C bond) |