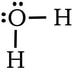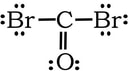ELectron Domains
VSEPR Theory
Now that we have established the different ways that molecules bond together using Lewis dot structures, we must talk about the limitations of them. Unfortunately, the bonding is only a component of what gives a compound its chemical and physical properties. The shape of the molecule also has a big influence on the properties of the compounds. Therefore, we must spend some time discussing the shapes of the molecules.
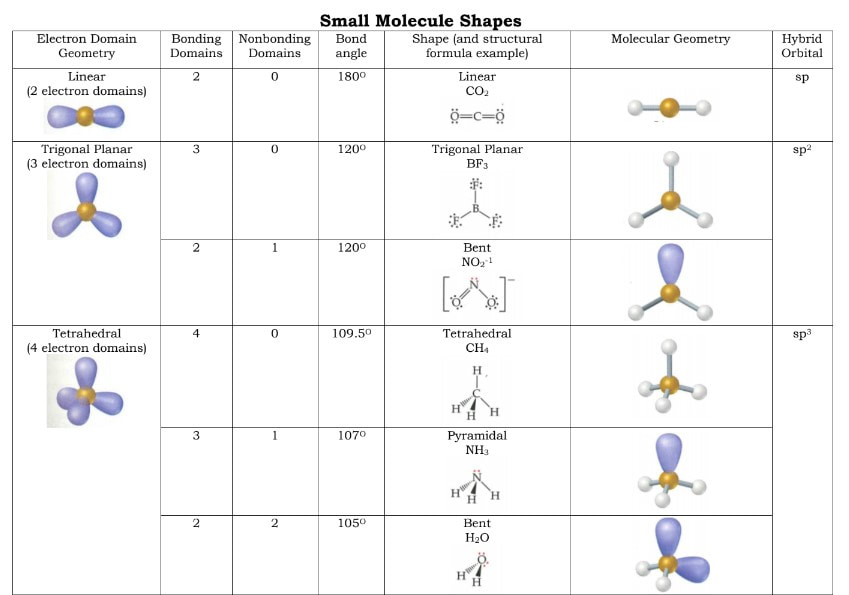
The molecular shape is largely due to the spacing of the bonds. The number of bonds and the number of electrons on the atoms will influence the shape due to the VSEPR Theory. VSEPR is an acronym that stands for Valence Shell Electron Pair Repulsion. This theory simply states that the valence shell electrons that are paired up (either as extra electrons on the atom or as a bond between two atoms) will repel one another to space themselves as far away from one another as possible. The number of pairs of electrons will directly influence the shape of the molecule due to this three dimensional spacing. And, because the Lewis dot structures are drawn in two dimensions, it is not the same as the shape that actually exists in three dimensions.
Electron Domains
In order to simplify things, we have developed this idea of electron domains that exist around the central atom of a molecule. An electron domain is a pair of electrons, either as a bond or as a pair of unshared electrons, on the central atom. These domains will help to determine which category of domain shape a molecule is in. To determine the number of electron domains, you have to count the number of bonding domains and the number of excess pairs of valence electrons on the central atom of the molecule. Here are a few examples:
Now that we have established the different ways that molecules bond together using Lewis dot structures, we must talk about the limitations of them. Unfortunately, the bonding is only a component of what gives a compound its chemical and physical properties. The shape of the molecule also has a big influence on the properties of the compounds. Therefore, we must spend some time discussing the shapes of the molecules.
The molecular shape is largely due to the spacing of the bonds. The number of bonds and the number of electrons on the atoms will influence the shape due to the VSEPR Theory. VSEPR is an acronym that stands for Valence Shell Electron Pair Repulsion. This theory simply states that the valence shell electrons that are paired up (either as extra electrons on the atom or as a bond between two atoms) will repel one another to space themselves as far away from one another as possible. The number of pairs of electrons will directly influence the shape of the molecule due to this three dimensional spacing. And, because the Lewis dot structures are drawn in two dimensions, it is not the same as the shape that actually exists in three dimensions.
Electron Domains
In order to simplify things, we have developed this idea of electron domains that exist around the central atom of a molecule. An electron domain is a pair of electrons, either as a bond or as a pair of unshared electrons, on the central atom. These domains will help to determine which category of domain shape a molecule is in. To determine the number of electron domains, you have to count the number of bonding domains and the number of excess pairs of valence electrons on the central atom of the molecule. Here are a few examples:
|
Example 1: H2O. Four domains. 2 bonding domains (O-H) and 2 nonbonding domains (extra pairs of electrons on O)
|
Example 2: COBr2. Three domains. One each for the C-Br bonds, and the double bond between C=O counts as one domain.
|
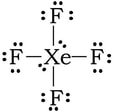
Example 3: XeF4. Six domains. 4 bonding domains (Xe-F) and 2 nonbonding domains (extra pairs of electrons on Xe)
|
|
Electron Domain Geometries
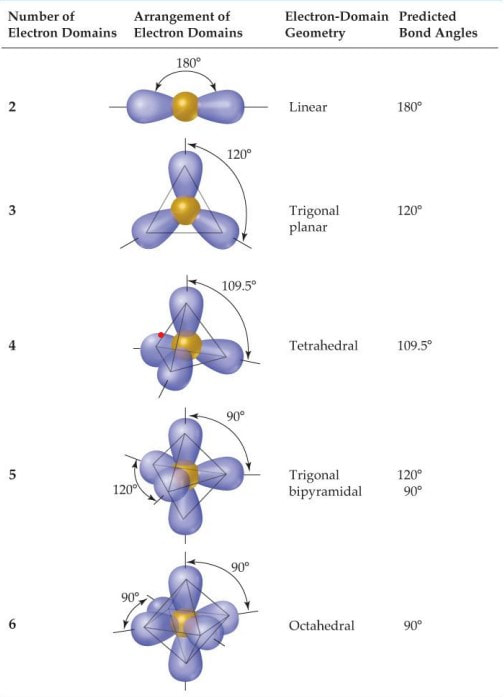
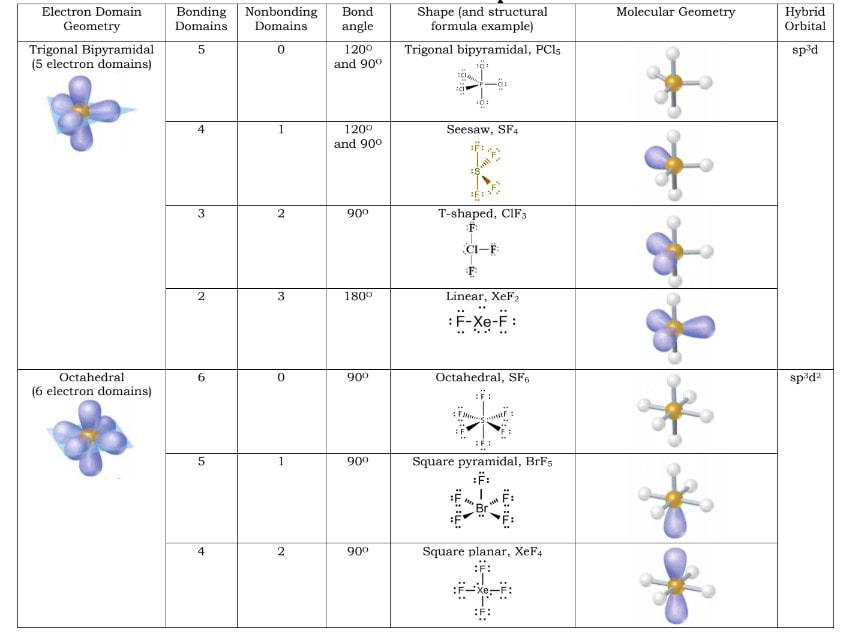
Electron domain geometries (or domain shapes) occur by getting the electrons as far away from one another as possible. Think of balloons. Balloons are going to position themselves as far apart from one another as possible. The more balloons you hold together by their tie off points, the closer they are and the arrangement in three dimensions change. There are 5 domain geometries. They are listed below and pictures of them are to the right. # of Domains Domain shape 2 Linear 3 Trigonal Planar 4 Tetrahedral 5 Trigonal Bipyramidal 6 Octahedral It does not matter if the domains are bonding or nonbonding domains when determining the domain geometries. However, the actual shapes of the molecules will be based off of these geometries, but will not necessarily be these shapes because of nonbonding geometries. What that means is, within these geometries, there are different shapes when there are all bonding domains versus when there are some bonding and some nonbonding domains. A further breakdown of the shapes is below: |