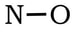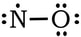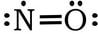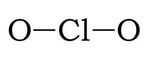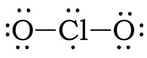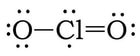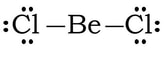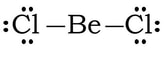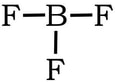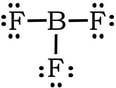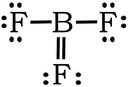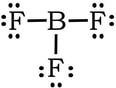Exceptions to Octet rule
.In covalent compounds, there are many molecules that do not follow the octet rule. They are still stable compounds, they just have one (or more) atoms within the molecule that do not have 8 valence electrons through sharing. There are three categories of compounds that do not obey the octet rule: Odd number of electrons, less than an octet, and more than an octet. Let's look at each of the three categories and see some examples.
Odd Number of Electrons
When the total number of valence electrons is an odd number, there is no way to achieve an octet on all atoms. In these cases, what we do is try to get all the outside atoms to an octet, and the central atom to 7 valence electrons. Here are a couple of examples:
Odd Number of Electrons
When the total number of valence electrons is an odd number, there is no way to achieve an octet on all atoms. In these cases, what we do is try to get all the outside atoms to an octet, and the central atom to 7 valence electrons. Here are a couple of examples:
|
Example 1: NO
1) Nitrogen has 5 valence electrons and oxygen has 6 valence electrons, so 5 + 6 = 11. 2) 3 & 4) We have 9 more electrons to place, so we finish the oxygen with 6, then add the last 3 electrons to the nitrogen:
5) The nitrogen currently only has 5 electrons. We can create a double bond between the N and O, keeping O at 8 valence electrons, and getting the N up to 7. In this instance, all of the formal charges are zero, so we will leave the N with one less than an octet:
|
Example 2: ClO2
1) Chlorine has 7 valence electrons and each oxygen has 6 valence electrons, so 7 + 2(6) = 19.
2) 3 & 4) We have 15 more electrons left. So, we add 6 electrons each to the oxygens, and the remaining 3 on the chlorine:
5) At this point, we have 7 electrons on the Cl. If we create two double bonds between the Cl the two Os, that will give Cl 9 VE. However, if we do this, there will be more atoms with a formal charge of zero (or closer to zero), so this is the most likely structure:
|
Less than an Octet
Some elements achieve less than an octet to be stable. The chief rule breakers are beryllium and boron. They covalently bond with other atoms and get to 4 and 6 valence electrons, respectively.
Some elements achieve less than an octet to be stable. The chief rule breakers are beryllium and boron. They covalently bond with other atoms and get to 4 and 6 valence electrons, respectively.
|
Example #3: BeCl2
1) Be has 2 valence electrons, each chlorine has 2, so 2 + 2(7) = 16 VE
2) 3) We complete the octet on the outside Cl.
4) We may be tempted to complete the octet on Be, by creating 2 double bonds.
However, we have a problem. In terms of electronegativity, chlorines electronegativity is so high comparted to beryllium, there is no way that Be is going to steal a set of electrons from Cl to form double bonds. Plus, the formal charges are all zero with single bonds, so we revert back to the first Lewis structure:
|
Example 4: BF3
1) B has 3 valence electrons, each fluorine has 7, so 3 + 3(7) = 24 VE
2) 3) Complete the octet on the fluorines.
We have no extra electrons, so we would be tempted to create a double bond between B and one of the Fs:
Same issue: In terms of electronegativity, fluorines electronegativity is so high comparted to boron, there is no way that B is going to steal a set of electrons from F to form a double bond. Plus, the formal charges are all zero with single bonds, so we revert back to the first Lewis structure:
|
More than an Octet
The largest category of rule breakers is the more than an octet group. These happen more often. What happens in this case is stable compounds form, and there are either too many electrons to make everything have an octet, or something bonds too many times, making more than an octet. So, we believe the central atom ends up with more than an octet of valence electrons.
The largest category of rule breakers is the more than an octet group. These happen more often. What happens in this case is stable compounds form, and there are either too many electrons to make everything have an octet, or something bonds too many times, making more than an octet. So, we believe the central atom ends up with more than an octet of valence electrons.
|
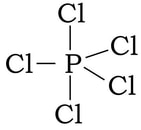
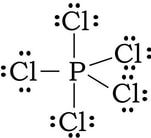
Example: PCl5
1) Phosphorous has 5 valence electrons, and each of the five chlorine have 7 valence electrons, so 5 + 5(7) = 40 electrons.
2) 3) At this point we have used 10 of the 40 electrons. Now we complete the octets on the chlorine by adding 6 dots (electrons) to each of the chlorine:
4) By doing this, we have used up all the electrons. But, at this point, the phosphorous has 10 VE, having bonded 5 times, We cannot make this work in any other way to satisfy the octet rule. And all formal charges work out to be zero, so we just stop here with more than an octet on the central atom.
|
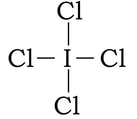
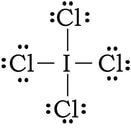
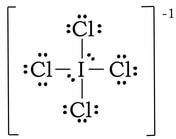
Example: ICl4-1
1) Iodine has 7 valence electrons, each of the chlorine have 7 and with a negative charge, there is one extra electron. So 7 + 4(7) + 1 = 36 electrons.
2) 3) To complete the octet on the outer chlorines, we add 6 electrons to each:
4 & 5) At this point we have used 32 of our 36 VE. Right now, everything has an octet, but we have extra electrons. If we place the leftover electrons on the I in the center, the I will have at total of 12 VE. Since there is no other way we can do this, and all of the formal charges are as close to zero as we can make them, we finish with this:
|