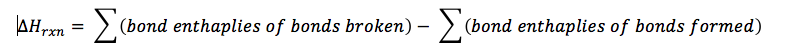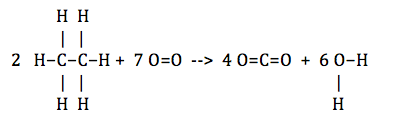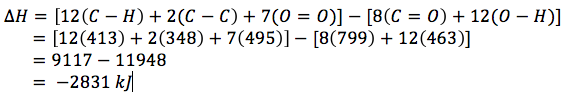Bond Length
In order to break a bond, energy is required. If a bond is formed, then energy has to be put in to break the bond. Conversely, to create a bond energy is released. This is measured as a bond enthalpy. The greater the bond enthalpy, the stronger the bond. Since this an enthalpy, it is given the symbol ΔH. Most textbooks have a list of bond enthalpies in them, so it is possible to determine the enthalpy of a chemical reaction by subtracting sum of all the bond enthalpies of bonds broken minus the sum of all the bond enthalpies of bonds formed, or
|
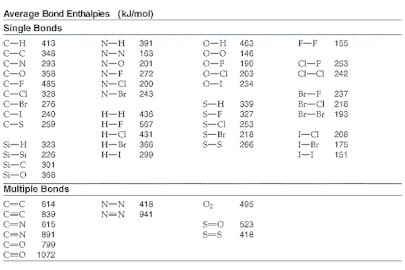
To the right is a table that lists typical bond enthalpies for some typical covalent bonds. Using these, we could determine the enthalpy of a reaction using the above equation.
Example: Determine the ΔHrxn for the combustion of ethane.
First, we need to write the reaction, and more importantly, determine the bonds that need to be broken and formed. The reaction is: 2 C2H6 (g) + 7 O2 (g) --> 4 CO2 (g) + 6 H2O (g)
To see the bonds, it may be better to draw it like this:
|
So, to count, there are 12 C-H bonds, 2 C-C bonds, 7 O=O bonds, 8 C=O bonds and 12 O-H bonds. The calculation for the enthalpy is then
|
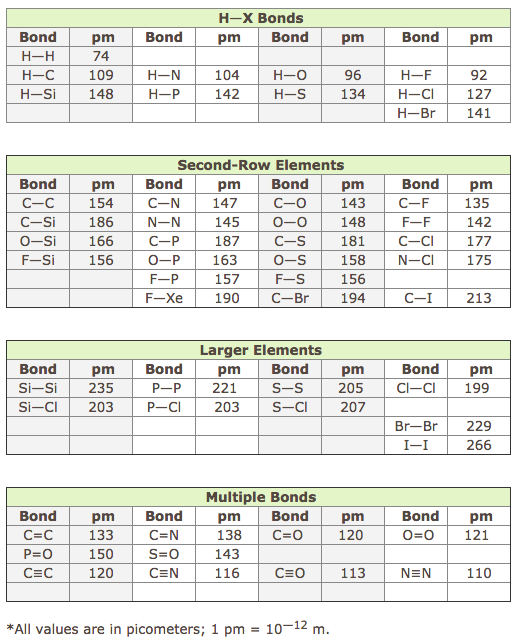
Bond Length
Related to bond enthalpy, we can look at bond length. Bond length is the average length of a particular type of bond. A table is to the right. Of particular interest is the relationship to bond length to enthalpy. It is no coincidence that the greater the bond length, the lower the bond enthalpy. If the atoms do not take much to break the bonds, then they are probably not close together. In addition, if you notice the bond length of a C-C single bond, it is longer than a C=C double bond. The more connections between two atoms, the shorter the bond length. As the number of bonds between atoms increase, the bond enthalpy will increase and the bond will become shorter. |