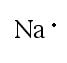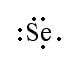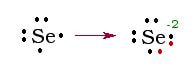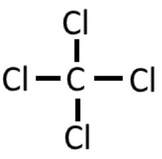Lewis Dot Structures
|
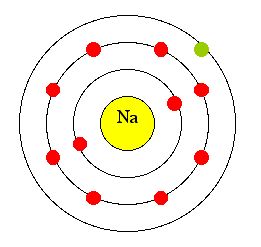
Lewis dot structures are used to show how the atoms of a compound bond together. Bonding occurs between the valence electrons, electrons in the outermost level of the atom. As an example we can look at this picture of the electron energy levels of sodium:
Sodium has 11 electrons, 2 electrons in the first energy level (orbit), 8 electrons in the second, and 1 in the third. Since there is one electron in the outermost level, sodium has one valence electron. The valence electrons follow a pattern similar to the charges and the electron configurations on the periodic table: |
|
Group Number (Name)
Group 1 (alkali metals) Group 2 (alkali earth metals) Group 13 (boron group) Group 14 (carbon group) Group 15 (nitrogen group) Group 16 (oxygen group) Group 17 (halogens) Group 18 (noble gases) |
Number of valence electrons
1 2 3 4 5 6 7 8 |
A Lewis dot structure for an element shows two things: the symbol with an appropriate number of dots representing the proper number of valence electrons.
Examples:
Examples:
The standard way of dot placement in a Lewis dot structure has an electron placed in the four directions (North, South, East and West), and then when more than 4 are needed listed, the electrons begin pairing up:
Octet Rule
Atoms tend to gain, lose or share electrons in order to acquire a full set of valence electrons. All atoms want to be stable, so they wish to achieve a stable electron configuration, like the noble gas. Look at the following examples:
Atoms tend to gain, lose or share electrons in order to acquire a full set of valence electrons. All atoms want to be stable, so they wish to achieve a stable electron configuration, like the noble gas. Look at the following examples:
|
Example 1: Selenium has 34 electrons. In order to become stable, selenium must obtain the same configuration as xenon. To do this, selenium gains 2 electrons. When this occurs, the addition of 2 electrons brings selenium’s total electrons up to 36. Now if we look at the valence electrons, by adding 2 electrons to the 6 valence electrons, selenium obtains 8 valence electrons.
Example 2: Sodium works much in the same way. The closest noble gas to sodium is neon. To achieve the same configuration as neon, sodium must lose one electron. By doing this, the outermost energy level becomes the 2nd energy level, which has 8 electrons. In both cases, the object is to get to 8 valence electrons. |
"Octet” comes from the idea that most atoms want to have eight electrons in its outermost level (shell). An octet is achieved by gaining, losing or sharing electrons.
An octet will contain 0 or 8 valence electrons. An octet is achieved with 0 valence electrons, because the next lower orbit becomes the outermost orbit, and this orbit will always have 8.
Exceptions to this rule are H, He and the transition metals. Helium is a noble gas and stable with 2 electrons. Hydrogen, having 1 electron, will desire to have the same configuration as helium, the closest noble gas. Therefore, hydrogen is also stable with 2 valence electrons. Transition metals can have varies levels of stability and various numbers of valence electrons, so as not to confuse us all, we’ll skip them for now.
An octet will contain 0 or 8 valence electrons. An octet is achieved with 0 valence electrons, because the next lower orbit becomes the outermost orbit, and this orbit will always have 8.
Exceptions to this rule are H, He and the transition metals. Helium is a noble gas and stable with 2 electrons. Hydrogen, having 1 electron, will desire to have the same configuration as helium, the closest noble gas. Therefore, hydrogen is also stable with 2 valence electrons. Transition metals can have varies levels of stability and various numbers of valence electrons, so as not to confuse us all, we’ll skip them for now.
Drawing Lewis Dot Structural Diagrams
When showing the bonding of covalent compounds, we follow a process:
1) Add up all the valence electrons for all atoms in the compound.
2) Write the symbols for the all atoms and connect them with a single bond. Typically the central atom is listed first, but not always.
3) Complete the octets of the atoms bonded to the central atom. Add enough dots to all the atoms so that with the 2 electrons in the bond, each of the outside atoms have 8 electrons.
4)Place any leftover electrons on the central atom. If after completing the octets on the outside atoms there are electrons left over from the total, add the extra electrons to the central atom.
5)If there are not enough electrons for the central atom to complete an octet on the central atom, try multiple bonds. Move pairs of electrons from the outside atoms to form another bond in the central atom. Do this as many times as necessary to complete the octet on the central atom.
Let's show a couple of examples worked out:
When showing the bonding of covalent compounds, we follow a process:
1) Add up all the valence electrons for all atoms in the compound.
2) Write the symbols for the all atoms and connect them with a single bond. Typically the central atom is listed first, but not always.
3) Complete the octets of the atoms bonded to the central atom. Add enough dots to all the atoms so that with the 2 electrons in the bond, each of the outside atoms have 8 electrons.
4)Place any leftover electrons on the central atom. If after completing the octets on the outside atoms there are electrons left over from the total, add the extra electrons to the central atom.
5)If there are not enough electrons for the central atom to complete an octet on the central atom, try multiple bonds. Move pairs of electrons from the outside atoms to form another bond in the central atom. Do this as many times as necessary to complete the octet on the central atom.
Let's show a couple of examples worked out:
|
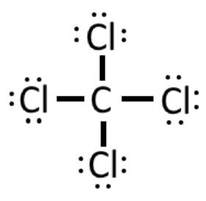
Example: CCl4
1) Carbon has 4 valence electrons, and each of the four chlorine have 7 valence electrons, so 4 + 4(7) = 32 electrons.
2) 3) At this point we have used 8 of the 32 electrons, as each bond counts as 2 electrons. Now we complete the octets on the carbon by adding 6 dots (electrons) to each of the chlorine:
4) By doing this, we have used up all the electrons. Fortunately, the C has an octet. Not only can chlorine claim the electrons in the bond, but so can carbon. As it is above, C and all 4 Cl have an octet. Steps 4 and 5 are not needed.
|
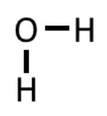
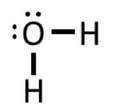
Example: H2O
1) Each of two two hydrogen has 1 electron, and oxygen has 6 valence electrons. So 2(1) + 6 = 8 electrons.
2) In this case, the O is the center atom, so 3) The hydrogen only need 2 electrons to be stable, which they already have in the drawing above, so we can skip step 3.
4) We have 4 more electrons from the 8 that we haven't used yet, so we put that on the central atom: 5) We have used all of our electrons, and the oxygen has an octet (4 electrons on the O, and then 2 bonds with 2 electrons each), so we can omit step 5.
|
|
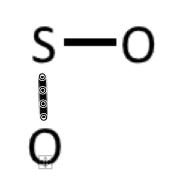
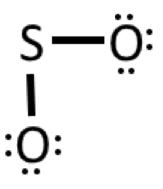
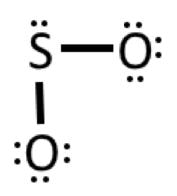
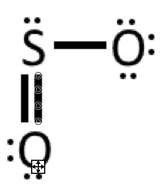
Example 3: SO2
1) Valence electrons: 6 + 2(6) = 18
2) Connect the atoms: 3) Complete octet on oxygens
4) Add 2 remaining electrons to central atom (sulfur)
5) The sulfur does not have an octet in the above diagram. We have no more electrons, so we must take some from the oxygen. If we take 2 electrons from the bottom electrons, and make them another bond between S and O, now both can have an octet. This is called a double bond.
|
Example 4: CO2
|