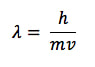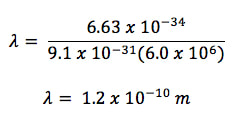Wave-particle duality
In the years following the Bohr model of the atom, many researchers investigated the nature of radiation. They found that, under the right conditions, radiation can not only interact with matter, it could also behave like matter. Conversely, certain tests on matter found that sometimes matter behaved like a wave. Through a process called X-ray diffraction, scientists proved that a stream of electrons move in a wave pattern, just like a stream of protons. French physicist Louis de Broglie predicted (and was later proven) that there is a relationship between the wavelength of matter as it moves to the momentum of the matter. In matter waves,
where λ is the wavelength, h is Planck's constant (6.63 x 10-34 J s), m is the mass (in kg) and v is velocity (in m/s).
Example 1: An electron with a mass of 9.1 x 10-31 kg is moving at a speed of 6.0 x 106. What is the wavelength of the electron?
All matter travels in waves according to this theory. It holds true because large objects (like the size of a person, or even a planet) will have such a large denominator that the wavelength will be so small it is not noticeable.
Heisenberg's Uncertainty Principle
Now that we know electrons can travels as waves, that means Bohr's model is not entirely true. Electrons that orbit around the nucleus will not be traveling around in a perfect circle, but in a wavy circle. Therefore, we do not exactly know where an electron is at any one moment. It could be a little closer to the nucleus, or a little bit further. Also, the electron is going so fast, that we cannot exact its position perfectly. The best we can do is say "about" where it is. The German physicist Werner Heisenberg proposed his uncertainty principle which says just this. We cannot determine the exact location of an electron, and know where it is going to go next. Or we might know where the electron is going next, but we do not know its exact position.
De Broglie and Heisenberg's work as described above furthers the idea that electrons travel around the nucleus, but not exactly the way Bohr envisioned. In fact, more recent discoveries suggest that although Bohr is right about the energy levels, the theory needs to include discreet areas where electrons can be found: orbitals.
Heisenberg's Uncertainty Principle
Now that we know electrons can travels as waves, that means Bohr's model is not entirely true. Electrons that orbit around the nucleus will not be traveling around in a perfect circle, but in a wavy circle. Therefore, we do not exactly know where an electron is at any one moment. It could be a little closer to the nucleus, or a little bit further. Also, the electron is going so fast, that we cannot exact its position perfectly. The best we can do is say "about" where it is. The German physicist Werner Heisenberg proposed his uncertainty principle which says just this. We cannot determine the exact location of an electron, and know where it is going to go next. Or we might know where the electron is going next, but we do not know its exact position.
De Broglie and Heisenberg's work as described above furthers the idea that electrons travel around the nucleus, but not exactly the way Bohr envisioned. In fact, more recent discoveries suggest that although Bohr is right about the energy levels, the theory needs to include discreet areas where electrons can be found: orbitals.