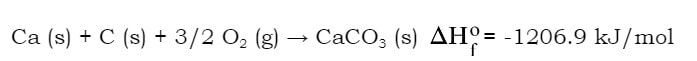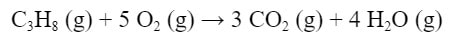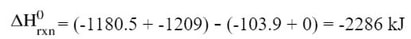Standard Enthalpy
|
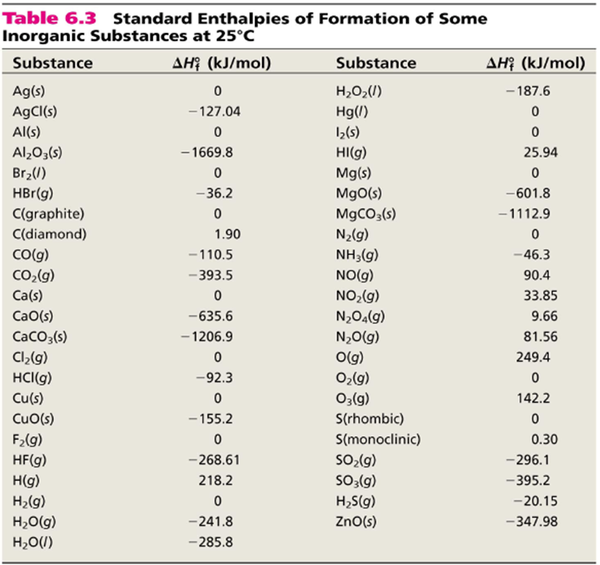
Standard enthalpy is the enthalpy of a reaction at 25 OC. It is measured in kJ/mol of the substance. For many substances, we know what the standard enthalpy of formation is for that substance. These numbers are usually listed in table form. For example, to the right is a small list of substances and their standard enthalpy of formations:
These values represent the amount of heat energy needed to form these materials from their elements. For instance, in the case of CaCO3, the equation to make calcium carbonate from its elements would be: A couple of things to notice here:
1) The compound is made from its elements in their natural form at 25 OC. 2) If you were to perform this reaction using the appropriate number of moles, you would release 1206.9 kJ of energy. 3) The standard enthalpies of formation of elements in their natural state are all 0 kJ/mol, because if you are making an element from its elements, it should take or release no energy. |
There is a more extensive list of standard enthalpies of formation in the back of the textbook in Appendix 4. There is also a bigger list at this link.
Using Standard Enthalpies of Formations to find Enthalpies of Reactions
The standard enthalpies of formation can be used to find the standard enthalpy of any reaction, as long as you know the enthalpies of the all the materials. For instance, say you have this reaction:
Using Standard Enthalpies of Formations to find Enthalpies of Reactions
The standard enthalpies of formation can be used to find the standard enthalpy of any reaction, as long as you know the enthalpies of the all the materials. For instance, say you have this reaction:
We can find the standard enthalpy of this reaction by taking the enthalpies of products minus the enthalpies of the reactants, or in mathematical terms:
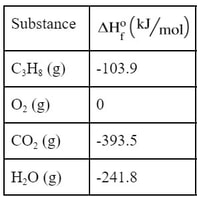
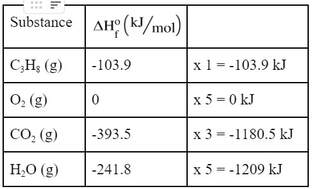
How do you use this? Well, the first thing you have to do is determine the ΔHO for each of the substances in the reaction. So, we go to the back of the book or online to find these standard enthalpies:
Now, these ΔHO are for one mole each. So, what we are going to have to do is multiply the ΔHO times the mole coefficients in the reaction:
Then we subtract the products minus the reactants: