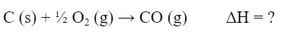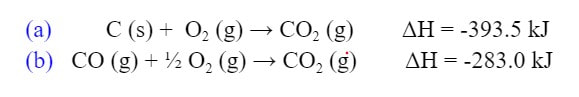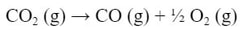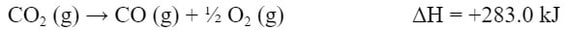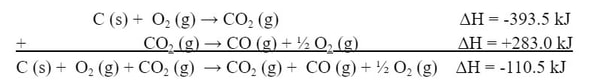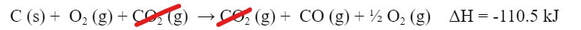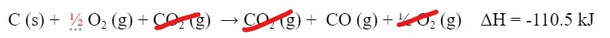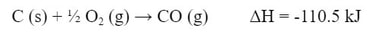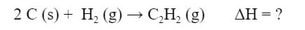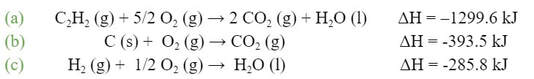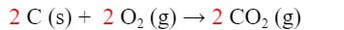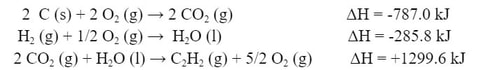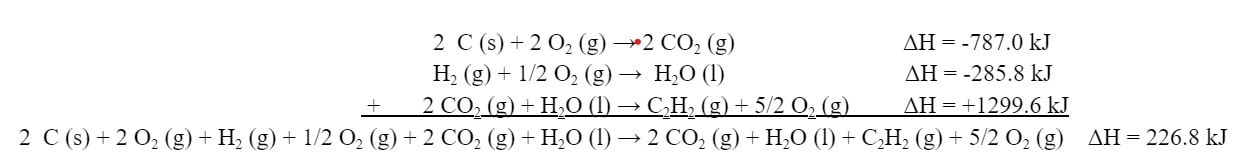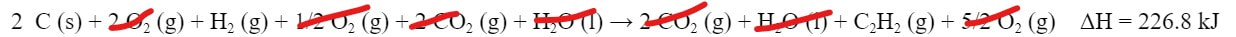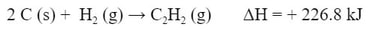Hess's Law
Hess's Law is one of the most used and most difficult to explain in words of the ideas in chemistry. The original law, in modern English, states that if a reaction is carried out in a series of steps, the enthalpy of the overall reaction equals the sum of the enthalpies of the individual reactions. What that really means is that if you knew the ΔH of several chemical reactions, and you wanted to determine the ΔH of another reaction, as long as the several chemical reactions add up to your final reaction, the sum of the ΔHs of the reactions is equal to the new reaction you are trying to get. Confused yet? Well, this law is easier to see if you do an example:
Example #1: Determine the enthalpy of the reaction
knowing the enthalpy of the following reactions:
In order to solve this, we need to construct the reaction we want from the reactions given. In these cases, you are rarely given a reaction you will not use. So, in order to get the C (s) we need, we will use the first reaction (a):
Since the O2 is in both reactions, let's move on to the CO (g). Now the CO (g) is in the second reaction (b). However, the CO (g) is on the wrong side of the reaction. So we need to flip the reaction around:
Because the products are now the reactants, the reaction is now endothermic, not exothermic. So, the ΔH is now positive:
Now, we can add the two reactions together, including the enthalpies:
The above reaction is not exactly what we wanted. Our goal reaction was different. We don't really want the CO2 in the reaction, and we have too much O2. However, much like net ionic equations, if something appears on both sides of the reaction, it's not really doing anything, and we can remove it. So, the CO2 can come out:
And 1/2 O2 can come out:
And, if we take those things out, we are left with the reaction we want.
Here's another example:
Example #2: Calculate the ΔH for the reaction:
given the following chemical equations and their respective enthalpies:
So, we need to assemble reaction (1) from the reactions given. We need 2 C (s), and we can get C (s) from reaction (b). But we need TWO C (s), not one. We will therefore double the reaction by multiplying the coefficients all by 2:
Because we doubled the moles of reactants, there will be twice as much heat involved. So, we will need to double the ΔH as well:
For the H2, we can use reaction (c):
C2H2 is available in reaction (a). However, it is on the wrong side. We will need to flip the reaction around, and change the sign of the ΔH:
Now we can add the reactions together, along with the ΔHs:
And then eliminate anything that is on both sides of the reaction:
Thus, getting this reaction: