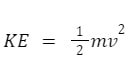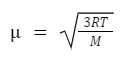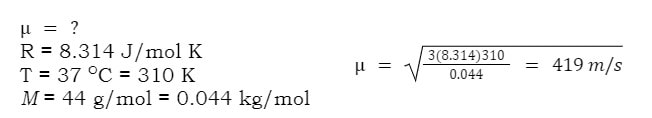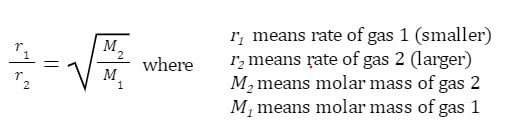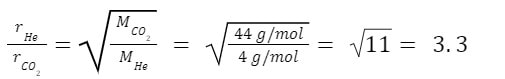Applications of The Kinetic Molecular theory
|
The Kinetic Molecular Theory
The kinetic molecular theory is the theory that within gases, all the molecules are moving. The theory has several postulates that are apart of it. They are:
|
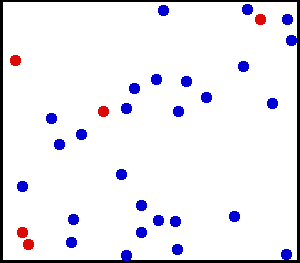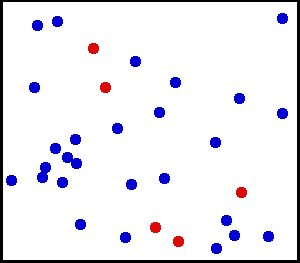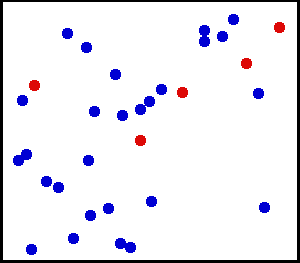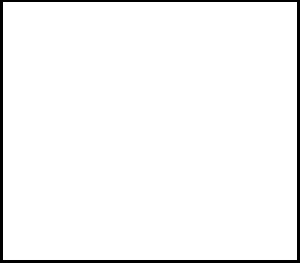
For these rules of the theory, many animations have been produced to show these. These simulations attempt to include, as best possible, the properties of these postulates. One such animation is shown above. Notice that in the animation, the particles motion appears to be random. Particles collisions can slow one particle down, but the other moves faster, thus having no net force lost in the collision.
Root Mean Square Speed
Temperature is a measure of the kinetic energy of the particles in a substance. At any given temperature, that means that the average kinetic energy of a gas particle is identical to any others. This means that if you have helium and carbon dioxide in the same tank, under the same kinetic energy, the gas particles will have the same kinetic energy. But that does not mean they will have the same speed of motion. Kinetic energy is calculated through the equation
Temperature is a measure of the kinetic energy of the particles in a substance. At any given temperature, that means that the average kinetic energy of a gas particle is identical to any others. This means that if you have helium and carbon dioxide in the same tank, under the same kinetic energy, the gas particles will have the same kinetic energy. But that does not mean they will have the same speed of motion. Kinetic energy is calculated through the equation
Where m is mass and v is velocity (or speed). If the KE of the helium is the same as the carbon dioxide, because carbon dioxide is a heavier molecule (44 amu) than helium (4 amu), the velocity of the helium molecule must be considerably faster than the carbon dioxide. By combining the KE equation with the ideal gas law, it is possible to determine the root mean square speed of any gas molecule under certain temperature conditions:
In this equation, μ stands for rms speed, R is the gas constant, T is temperature and M is the molar mass. However, for this equation to work, R must be 8.314 J/mol K, and M must be in kg/mol.
Example: What is the rms speed of carbon dioxide exhaled from the human body (37 OC)
In order to do this calculation we must determine the values of all the components of the equation:
Effusion
Effusion is a measure of how fast a gas will escape from a small hole within a container. As you may know, a helium balloon seems to shrink and lose its "air" faster than a regular balloon. That is because the helium molecules is smaller and faster than the regular "air" molecules. The fact that the helium is smaller means it can more easily fit out of the small, microscopic holes that tear in the latex balloon over time. And, since the helium is also going faster, it is more likely to hit the hole more often. Therefore, the smaller the molar mass (size) of the gas molecule, the faster it will effuse. Determining how fast a gas can effuse from a hole is beyond the scope of this course. This calculation involves hole size, temperature, molar mass, molecule radius, and other things.
However, using something called Graham's law, we can determine how much faster a small molecule can effuse out of a hole compared to a larger molecule. This equation is represented:
Effusion is a measure of how fast a gas will escape from a small hole within a container. As you may know, a helium balloon seems to shrink and lose its "air" faster than a regular balloon. That is because the helium molecules is smaller and faster than the regular "air" molecules. The fact that the helium is smaller means it can more easily fit out of the small, microscopic holes that tear in the latex balloon over time. And, since the helium is also going faster, it is more likely to hit the hole more often. Therefore, the smaller the molar mass (size) of the gas molecule, the faster it will effuse. Determining how fast a gas can effuse from a hole is beyond the scope of this course. This calculation involves hole size, temperature, molar mass, molecule radius, and other things.
However, using something called Graham's law, we can determine how much faster a small molecule can effuse out of a hole compared to a larger molecule. This equation is represented:
Example: How much faster does helium effuse from a hole compared to carbon dioxide?
Answer:
Answer:
Essentially, what this means is that helium will effuse 3.3 times faster than the carbon dioxide, under the same conditions.
Diffusion
Diffusion is the idea that one gas should be able to move through space or another gas based on the random motion of the particles. There are calculations for this, but they are beyond the scope of this course. However, you should know that the following factors affect diffusion:
Diffusion
Diffusion is the idea that one gas should be able to move through space or another gas based on the random motion of the particles. There are calculations for this, but they are beyond the scope of this course. However, you should know that the following factors affect diffusion:
- the smaller the molecules, the faster the diffusion rate will be
- the higher the pressure, the slower the diffusion rate will be
- the higher the temperature, the faster the diffusion rate will be