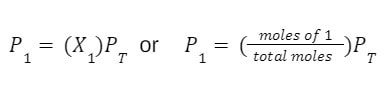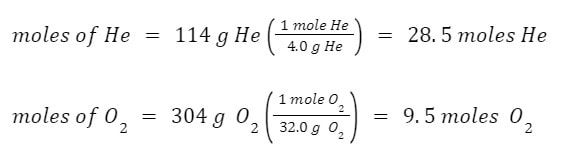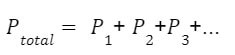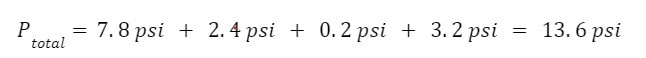Partial Pressures
|
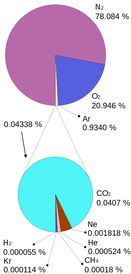
Often, when you are dealing with a gas (in regards to the gas laws), the gas is a mixture of several different gases. For instance, air is a mixture of several different gases: nitrogen is the most abundant, at roughly 78% by mole fraction. Then, oxygen is second at 21%. Argon, carbon dioxide, water vapor and other trace gases and pollutants make up the last 1%. The pie chart to the left gives a representation, courtesy of Wikipedia.
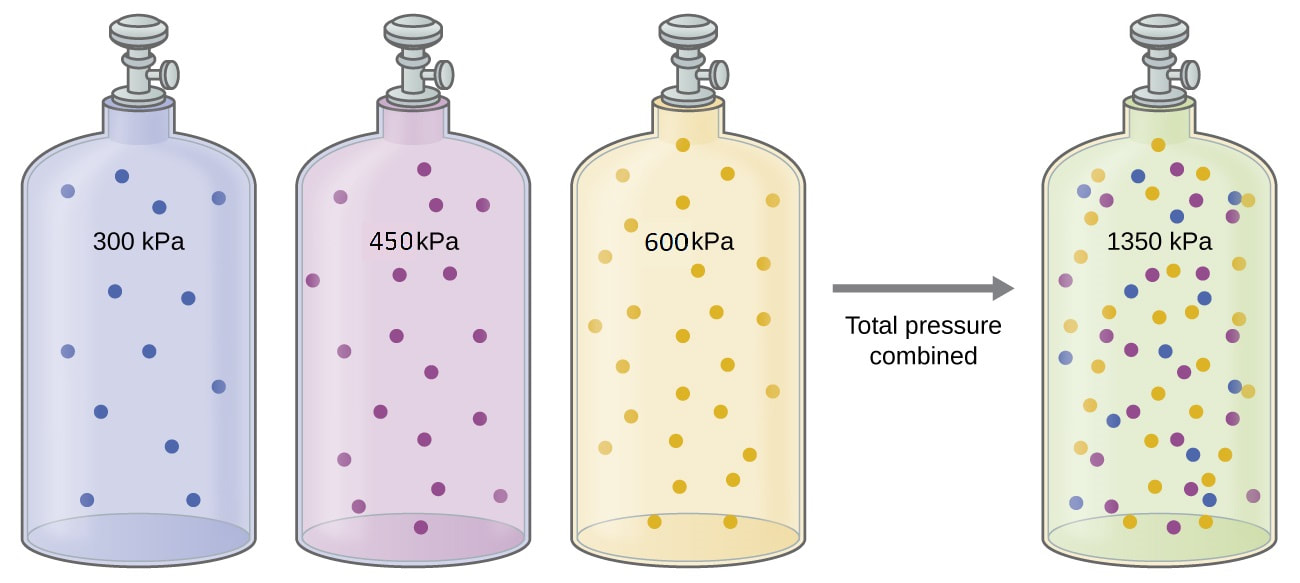
When you have a mixture of gases, each gas in the container collides with the walls of the container. This exerts a pressure. Therefore, every gas in the container exerts its own pressure. Each of these pressures are independent of the other pressures in the container. Therefore, since each gas exerts its own pressure, based on the number of particles it has (mole fraction), we can determine these pressures using the ideal gas law. We call the individual pressures a partial pressure. So, in air, since the air has a pressure of 1.00 atm, and the mole fraction of the nitrogen is 78%, the partial pressure of the nitrogen in the air is 0.78 atm. |
Calculating Partial Pressures and Total Pressures
There are really two applications of this idea that you will need to be able to calculate. You may be given the total pressure and asked to find the partial pressures, or have the partial pressures and need to find the total pressures. Let's look at each calculation:
First, if you have the total pressure of the mixture, and need to determine the partial pressure of one of the gases in the mixture, you will need the mole fraction (or mole percentage) of the gas in question and the total pressure of the mixture. In this case you can use the following manipulation of Dalton's Law of Partial Pressures:
There are really two applications of this idea that you will need to be able to calculate. You may be given the total pressure and asked to find the partial pressures, or have the partial pressures and need to find the total pressures. Let's look at each calculation:
First, if you have the total pressure of the mixture, and need to determine the partial pressure of one of the gases in the mixture, you will need the mole fraction (or mole percentage) of the gas in question and the total pressure of the mixture. In this case you can use the following manipulation of Dalton's Law of Partial Pressures:
Example: A SCUBA tank is holding 114 grams of helium and 304 grams of oxygen, by mass. The total pressure in the tank is 17.0 atm. What is the partial pressure of the oxygen?
Answer: In order to find the partial pressure of the oxygen, we need to find the mole fraction of the oxygen. So, we will need the moles of both gases:
Answer: In order to find the partial pressure of the oxygen, we need to find the mole fraction of the oxygen. So, we will need the moles of both gases:
Then, we calculate the partial pressure of the oxygen by taking the total pressure of 17.0 atm times the mole fraction (percent) of oxygen in the mixture:
Example: A 5.0 L tank is filled with 4 gases. The partial pressure of the gases are 7.8 psi for nitrogen, 2.4 psi for oxygen, 0.2 psi for carbon dioxide and 3.2 psi for argon. What is the total pressure inside the tank?
Answer:
Answer: