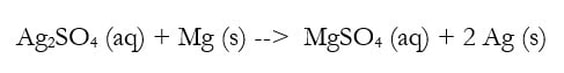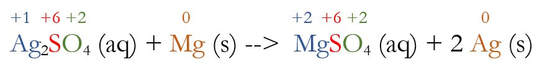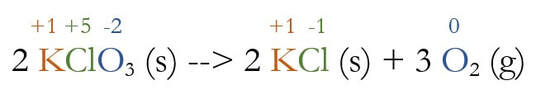Oxidation Numbers
Often, it is not easy to tell if a reaction is a redox reaction, let alone what element is being oxidized or reduced. In general, all synthesis, decomposition, single replacement and combustion reactions are redox reactions. However, it is difficult to tell which element is being oxidized or reduced. And which elements are doing neither. Scientists have developed element identification rules to determine this through oxidation numbers. Oxidation numbers are a lot like charges on an ion in a compound. However, the difference is an oxidation number can be assigned to every element in a compound, even if the element is part of a polyatomic ion group. These oxidation number rules are as follows:
Rules for Oxidation Numbers
1) An atom in its elemental form has a oxidation number of zero.
2) Alkali metals = +1. Alkali Earth metals = +2, Aluminum = +3
3) Oxygen: usually -2, except in peroxide (O2-2) where it is -1.
4) Hydrogen: +1 with a nonmetal, -1 with a metal
5) Halogens: -1 in a binary compound, when combined with oxygen in a polyatomic ion, the charge will be positive.
*6) Sum of oxidation numbers: For a neutral compound is zero. The sum of oxidation numbers for a polyatomic ion is equal to its charge.
2) Alkali metals = +1. Alkali Earth metals = +2, Aluminum = +3
3) Oxygen: usually -2, except in peroxide (O2-2) where it is -1.
4) Hydrogen: +1 with a nonmetal, -1 with a metal
5) Halogens: -1 in a binary compound, when combined with oxygen in a polyatomic ion, the charge will be positive.
*6) Sum of oxidation numbers: For a neutral compound is zero. The sum of oxidation numbers for a polyatomic ion is equal to its charge.
* As you can see, there are many elements that do not have a rule. If an element doesn't have a rule, it goes by rule 6. Rule 6 means that if an element doesn't have a rule, then all the oxidation numbers must add up to zero for a neutral compound. Since copper in CuF2 doesn't have a rule, the Cu must balance out the charge of the F2 to make it neutral. Since the two flourines have a total charge of -2 (each one is -1), the copper must have a +2 oxidation state
In the instance of a polyatomic ion, like NH4+, the N doesn't have a rule. But, there is a rule for H. Since H is with a nonmetal, H must be +1 each, making a total charge of +4. The total charge of the grouping (because it is an ion) has to be +1, so the N must have an oxidation state of -3.
In the instance of a polyatomic ion, like NH4+, the N doesn't have a rule. But, there is a rule for H. Since H is with a nonmetal, H must be +1 each, making a total charge of +4. The total charge of the grouping (because it is an ion) has to be +1, so the N must have an oxidation state of -3.
Here are a few examples and how the oxidation states are arrived at:
|
1) Determine the oxidation number for Cr in
|
K2CrO4
|
Based on the rules, K is an alkali metal, which means it has an oxidation number of +1. Since there are two, the total charge of all the K is +2. The O has an oxidation state of -2, and since there are four O's, the total charge of all the O's is -8. The Cr must therefore have enough charge to make the total charge equal to zero. Since there is already a +2 and a -8, then the Cr must have an oxidation state of +6.
|
2) Determine the oxidation number of S in
|
SO3-2
|
Rule #3 says that oxygen is -2, and there are 3 of them so the Os account for -6 altogether. Now, the whole compound has an overall charge of -2, so the S must be enough so that when you add it to -6, you still get -2. Therefore, S must be +4.
|
3) Determine the oxidation number of Br in
|
HBrO3
|
Again, rule #3 says oxygen is -2, and 3 mean a charge of -6. The H has a charge of +1 because it is paired up with nonmetals. So to make the compound neutral, Br must be +5, because (+1) +(+5) +(-6) = 0.
Many more examples:
|
|
Determining what is being oxidized and what is being reduced
Often, it is asked what substance is being oxidized or reduced in a chemical reaction. To do this, we must use our oxidation rules and determine the charge of every element within a chemical reaction. For instance take this example:
Often, it is asked what substance is being oxidized or reduced in a chemical reaction. To do this, we must use our oxidation rules and determine the charge of every element within a chemical reaction. For instance take this example:
To determine what is being oxidized or reduced, an easy way is to determine the oxidation number for each element. I typically list those numbers above. They are color coded to make it easier for you to see below:
You can cheat a little when you know what the charge of a polyatomic ion is. For instance, I figured S out because I knew sulfate has a -2 charge overall. If each of the 4 Os is -2, then sulfur has to be +6 in order for the ion to add to -2. Then, silver was easy to figure out. Also, notice I put the oxidation number of just one of that type of element above it. The two Ags have to add up to +2, which makes each one a +1.
The two elements that change charge are Ag and Mg. Ag goes from +1 on the left to 0 on the right. Since the charge goes down, it becomes more negative. This means Ag had to gain electrons, which is reduction (GER). Another way to think of it is Ag's charge went down, or was reduced.
Mg on the other hand, went from 0 to +2. The only way for atoms to become more positive is to lose negative electrons, and the loss of electrons is oxidation (LEO).
Another example:
The two elements that change charge are Ag and Mg. Ag goes from +1 on the left to 0 on the right. Since the charge goes down, it becomes more negative. This means Ag had to gain electrons, which is reduction (GER). Another way to think of it is Ag's charge went down, or was reduced.
Mg on the other hand, went from 0 to +2. The only way for atoms to become more positive is to lose negative electrons, and the loss of electrons is oxidation (LEO).
Another example:
So, we use the rules to determine the oxidation numbers for all elements:
Cl and O both change oxidation numbers. O goes from -2 to 0, which means a loss of electrons to become more positive. This is oxidation. Cl goes from +5 to -1, which is a gain of electrons to become more negative, or reduction.
A couple more examples:
A couple more examples:
|
|