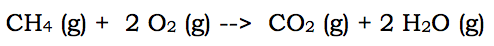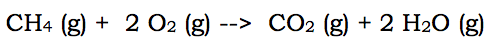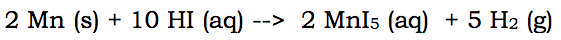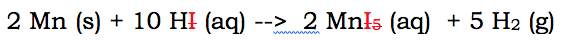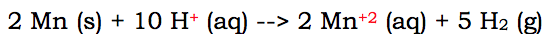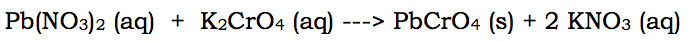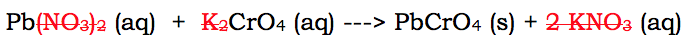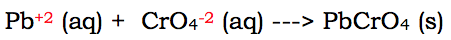Net Ionic Equation
When describing reactions, in AP Chemistry, the College Board has decided that the listing of reactions needs to be in net ionic form. This means that ions that do not actually change during a chemical reaction are omitted. Therefore, most (not all) reactions that you write need to be altered. In very simple terms, a net ionic reaction omits (leaves out) the spectator ions. Spectator ions are the aqueous ions that remain aqueous ions throughout the reaction. Let's break this down by using some examples of chemical reactions. Here is a combustion reaction:
The above reaction is the full reaction for the combustion of methane. To change a full reaction to a net ionic equation, it is necessary to remove any aqueous ions that appear the exact same way on both sides of the arrow. But, in the above reaction, there are no aqueous substances. Therefore, the full reaction and the net ionic reaction are the same.
For you to have to change a full reaction into a net ionic reaction, there needs to be some aqueous materials in the chemical reaction. Here's a single replacement reaction that needs to be altered:
Now this reaction has aqueous ions on both sides. Which means there are spectator ions. The spectator ion in this reaction is I. Iodine is aqueous on both sides of the reaction. Since it doesn't change states, and remains a negative I ion, it can be removed. It is removed because it doesn't really change.
If we remove the iodine, the ions the I is paired up with on both sides remain. However, they are not neutral because the negative I has been removed. Therefore, you must put the charge on the ions that remain. The final net ionic reaction looks like this:
Here's another example, a precipitation reaction:
In this reaction, there are two ions that remain aqueous throughout the reaction, the potassium and the nitrate. They are aqueous individually and together. This means we can remove them both:
When we remove these ions, the ions they were paired with (the lead and the chromate) will now be charged. Therefore, we must include their charges: