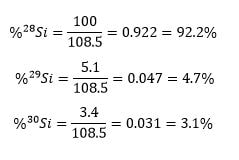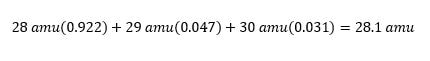Mass Spectroscopy
|
Determining Isotopes
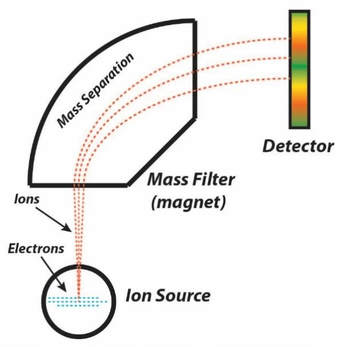
The number of isotopes of an element is determined by a mass spectrometer. In a mass spectrometer, an element is placed in a container where all the electrons are removed from the element. Once all the electrons are removed, the individual atoms of the same elements all have the same charge. They will all be attracted to a magnet in the same way. However, some will have a greater mass than others. As the element is shot upward in the diagram, it goes into the mass filter, where a magnet will attract the ions. Since the ions are moving at a high speed, they will be thrown off course by the magnet. The smaller the mass, the more it will be thrown off course by the magnet. Because of this, the larger mass isotopes will hit the detector in a different place than the smaller mass isotopes. The raw number of each kind of isotope that hits the detector is read off by a computer, and then the percentage of each mass is displayed in graphical form. The mass of each isotope can be calculated by where it hits the detector based off of mathematics |
|
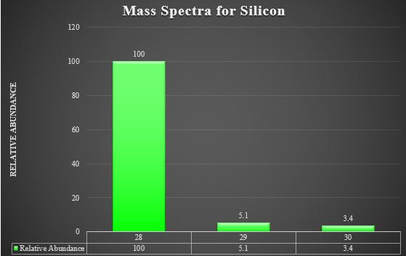
The data from the machine is then presented in graphical form similar to this. To the left is a mass spectra for silicon. According to the graph, there are three isotopes of silicon: Silicon-28, Silicon-29 and Silicon-30. Silicon-28 is in largest abundance, as that isotope has the highest relative abundance. The isotope in greatest abundance will typically be presented at 100, and all the rest will be a factor of that largest one. In this case, the total amount of silicon of all the isotopes is 100 + 5.1 + 3.4 = 108.5. We can find the percent abundance of each one by taking the relative abundance of each one and dividing by the total, in this case 108.5.
|
Using this data, we can use the masses and the abundances to determine the average atomic mass of the element:
Here's another explanation of how a mass spectrometer works.