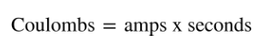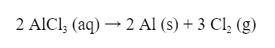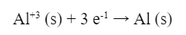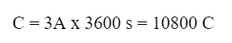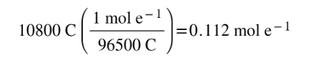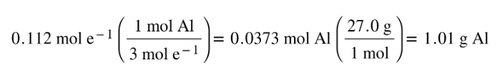Current and Stoichiometry
|
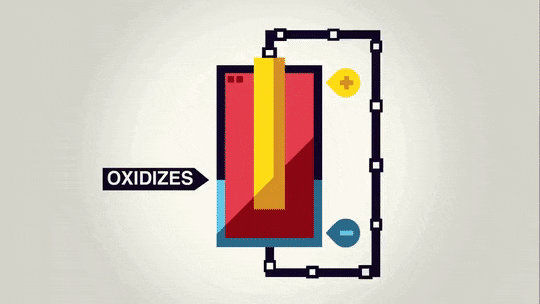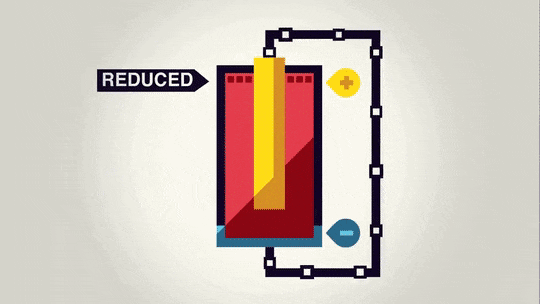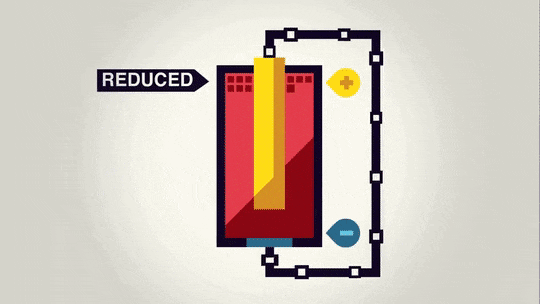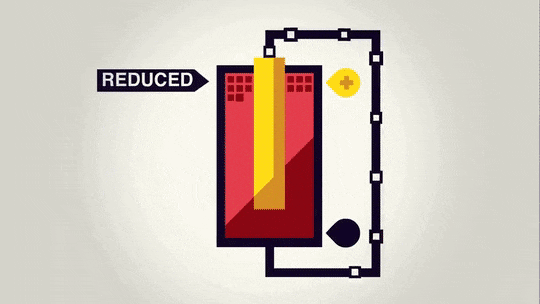
Now that we have spent a lot of time talking about the "power" of a battery, we should talk about what that power does. Essentially, the voltage is a measure of how much the electrons want to move from the anode to the cathode.
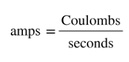
As we measure the flow of electrons, that is a measure of current in a unit called amps. Current is a measure of the amount of charge (measured in Coulombs) of electrons that flow past a point in the wire in a second, or: |
Michael Faraday, the creator of the electric motor, determined the amount of electrons in a Coulomb. The electrical charge of 1 mole of electrons is 96,500 C. Knowing these relationships of current and charge and number of electrons, we can use the information to stoichiometrically determine the amount of current to produce a certain amount of material in the process of electrolysis.
Often it is possible to decompose an ionic compound by running electricity through it. The electrons running through it move the electrons from one material to the other and cause a certain amount of material to be made depending on the size of the current and how long the current runs.
Example: Aluminum chloride can be decomposed by electrolysis of the solid. If a 3.0 A current is run through aluminum chloride for an hour, how many grams of aluminum will be created?
Solve: First we need a reaction for the decomposition:
Often it is possible to decompose an ionic compound by running electricity through it. The electrons running through it move the electrons from one material to the other and cause a certain amount of material to be made depending on the size of the current and how long the current runs.
Example: Aluminum chloride can be decomposed by electrolysis of the solid. If a 3.0 A current is run through aluminum chloride for an hour, how many grams of aluminum will be created?
Solve: First we need a reaction for the decomposition:
Then, we can separate the aluminum into a half reaction:
Now, we calculate the charge caused by the current:
Since 1 mole of electrons is equal to 96,500 C, then:
From the moles of electrons, we can then use the balanced equation to complete the stoichiometry to grams of aluminum: