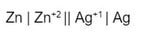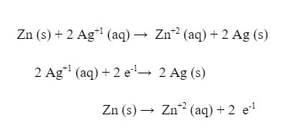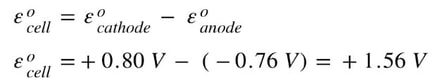Voltaic Cells
|
When we but batteries, the batteries have letters live AA, AAA, D, C and 9V to indicate the device they go in. These are actually indications to make it easy to know what kind of battery will fit in a give device. For instance, D, AA and AAA batteries all have the same "strength", just different sizes for use and longevity. When a battery gets made, the "strength" within the battery depends on the chemicals used in them. If you hook a device up to a 9V battery when it only uses one or two AA batteries, you'll likely fry the circuitry within.
So how do chemists measure the "strength" of the battery? |
Volts
When building a voltaic cell, there is a potential difference between where the electrons are (anode/negative end) and where they are attracted to (cathode/positive end). Potential difference is a fancy way of saying "how badly/quickly do the electrons want to move to the other end of the battery?" This has a real measurement. It's called voltage, or volts. Volts is a measure of the amount of energy that the electrons have per unit of charge, or
When building a voltaic cell, there is a potential difference between where the electrons are (anode/negative end) and where they are attracted to (cathode/positive end). Potential difference is a fancy way of saying "how badly/quickly do the electrons want to move to the other end of the battery?" This has a real measurement. It's called voltage, or volts. Volts is a measure of the amount of energy that the electrons have per unit of charge, or
|
This will not be a calculation we will be doing. I simply bring this up so that you know that volts measures the amount of energy (J) that a battery delivers per unit of charge. The greater the voltage, the more "strength" the battery has. You can change the volts by using different combinations of chemicals in your battery. Higher voltages occur when you have a substance with a lot of electrons that really doesn't want them on the anode and a cathode that really likes getting electrons.
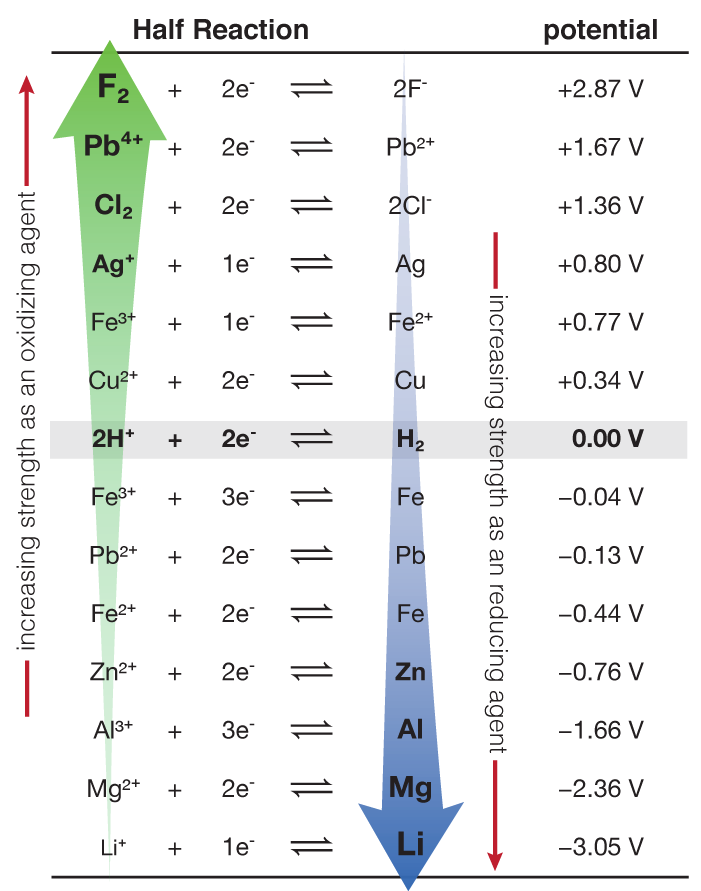
To calculate the voltage in a voltaic cell (battery), we use a table called a "Standard Reduction Potential". A brief version of the table (they can be much longer) is to the right. These are measured amount of voltage produced by different reduction half reactions. Think of them as "this is how much strength this particular reduction (gaining of electrons) has". Let's say you want to make a battery with silver and zinc. The voltaic cell representation would look like this: What this is saying is that Zn is acting as the anode, losing electrons to become Zn+2, while on the other side of the bridge, the Ag+1 is gaining the electrons to become Ag. Ag is the cathode. The reaction (and half reactions) would look like this:
|
When we split the reaction we see two half reactions and find that reduction is happening with Ag and oxidation is happening with Zn. We have already learned that the oxidation happens at the anode, and reduction happens at the cathode. To determine the voltage of the voltaic cell, we use the following equation:
Essentially, what we do is subtract the standard reduction potential listed in the table that happens at the cathode (reduction) minus the table value for the anode (oxidation). Since Ag is reduced and Zn is oxidized, we subtract the value for Ag from the table above minus the value for zinc. Or:
Couple of things of importance here:
- You do not need to figure in molar ratios.
- You have to identify what is being oxidized and what is being reduced. If you get a negative number, that simply means that the battery is being recharged, as you are moving the electrons back to the anode.
- There are many ways to ask to find the voltage. If the question asks for you to determine the voltage, the cell potential, the potential difference, the electromotive force or the EMF, all of these things mean find the voltage.