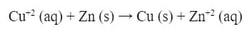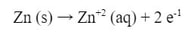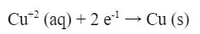Voltaic Cells
|
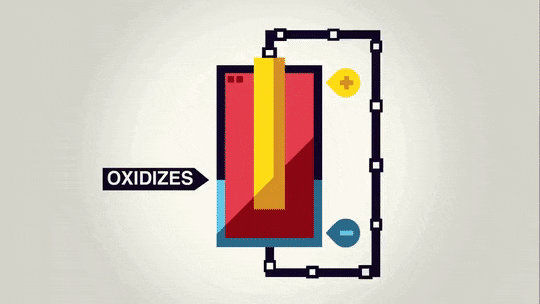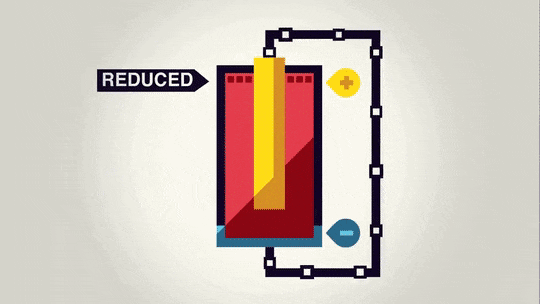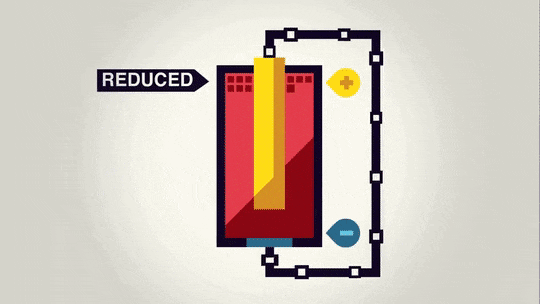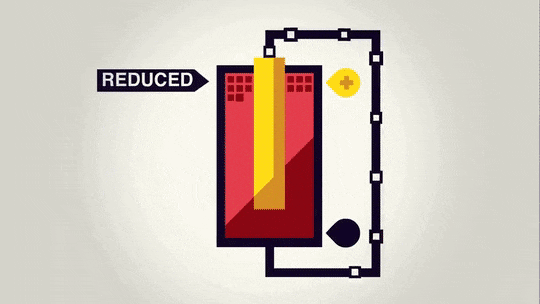
In redox reactions, electrons get moved from one substance from another as one substance loses electrons as they travel over to the other substance. The way batteries work is that the electrons move from one end of the battery to the other through a wire as the redox reaction occurs. In a battery, there is a chemical reaction happening at each end of the battery: Oxidation is happening at the negative end of the battery, and reduction is happening at the positive end of the battery. The electrons get transferred from the negative end of the battery to the positive end of the battery through a wire.
So, how does this happen? |
Voltaic Cells
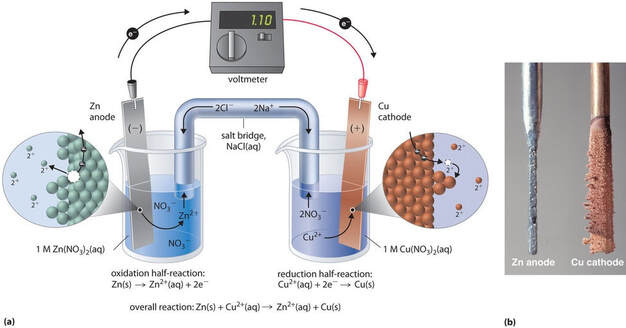
A battery scientifically is called a voltaic cell. In a voltaic cell, the chemical reaction does not happen by the two chemicals involved in the redox reaction coming in contact with one another. Rather, the oxidation and reduction are separated from one another. In order to make a voltaic cell, you have to have two beakers. One beaker has a metal that gives up electrons, in a solution with the same metal dissolved. Then, in another beaker, you have a metal that will gain electrons in a solution of that same metal. That's confusing. Let's talk about it using an example.
A battery scientifically is called a voltaic cell. In a voltaic cell, the chemical reaction does not happen by the two chemicals involved in the redox reaction coming in contact with one another. Rather, the oxidation and reduction are separated from one another. In order to make a voltaic cell, you have to have two beakers. One beaker has a metal that gives up electrons, in a solution with the same metal dissolved. Then, in another beaker, you have a metal that will gain electrons in a solution of that same metal. That's confusing. Let's talk about it using an example.
|
Let's say we are doing this reaction:
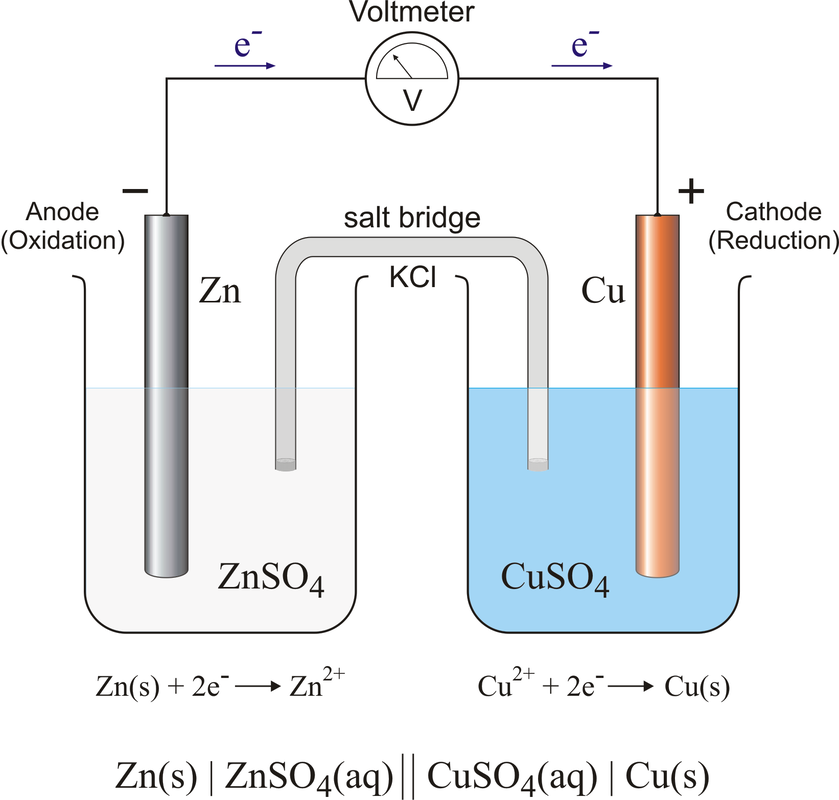
What you would do is have a piece of zinc in a solution of zinc, say zinc (II) sulfate. That would be in one beaker. Then, in a separate beaker, you would have a piece of copper in a copper solution, say copper (II) sulfate. (look to the right).
Now, initially nothing would happen. The zinc doesn't react with the zinc solution and likewise for the copper. However, if you connect a wire from the zinc metal piece to the copper metal piece, the reaction will start. At the zinc electrode (metal piece), electrons will leave the zinc as this reaction happens: The Zn starts to dissolve in Zn+2 ions that go into the solution, while the electrons travel through the wire to the Cu electrode. At the Cu electrode, the following reaction happens:
|
The Cu+2 ions in the CuSO4 solution start adding on to the copper piece of metal. The electrons attract the Cu+2 ions from the solution and start making the copper electrode bigger.
In this bigger picture, you can see that the zinc electrode is actually getting smaller as it "dissolves" into the solution. The copper electrode is actually growing in size as the electrons are combining with the copper ions in the solution to "precipitate" onto the copper metal piece.
The important part of this is actually the flow of electrons through the wire. Electricity runs almost all of our powered devices. Electricity is a flow of electrons through a wire. Through this simple setup, we can cause electrons to flow through the wire to power a device, all due to a simple reaction between two fairly common materials.
The important part of this is actually the flow of electrons through the wire. Electricity runs almost all of our powered devices. Electricity is a flow of electrons through a wire. Through this simple setup, we can cause electrons to flow through the wire to power a device, all due to a simple reaction between two fairly common materials.
The one thing not mentioned yet is the salt bridge in the middle. The salt bridge is usually a salt water solution, either NaCl or KCl, which provides ions to both solutions. As Cu+2 ions leave the CuSO4 solution, the solution becomes negatively charged due to the absence of Cu+2 ions. The salt bridge has positive and negative ions in it, and as the solution becomes more negative, positive ions from the salt bridge leave the bridge and go into the negative solution to keep it neutral. Similarly, the excess of Zn+2 ions in the other solution will draw negative ions from the salt bridge into the positive solution to keep it neutral as well. This completes the circuit of charge, so the reaction keeps going until the zinc electrode is gone (ie, the battery "dies")
Let's talk terminology:
Electrode - the pieces of metals in the solution.
Anode - the electrode where oxidation occurs. This is the electrode that loses electrons. It is the negative end of the battery (source of negative electrons).
Cathode - the electrode where reduction occurs. This the electrode that gains electrons. It is the positive end of the battery (electrons are attracted to the positive end).
Voltage - a measure of how much pull there is on electrons from one end of the battery to another.
Electrode - the pieces of metals in the solution.
Anode - the electrode where oxidation occurs. This is the electrode that loses electrons. It is the negative end of the battery (source of negative electrons).
Cathode - the electrode where reduction occurs. This the electrode that gains electrons. It is the positive end of the battery (electrons are attracted to the positive end).
Voltage - a measure of how much pull there is on electrons from one end of the battery to another.