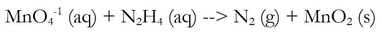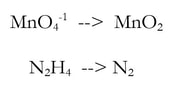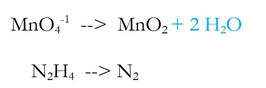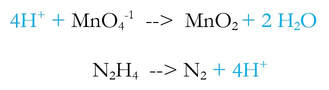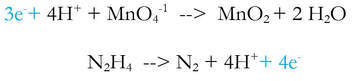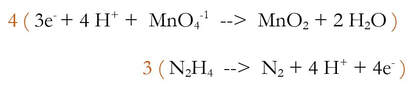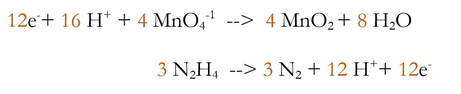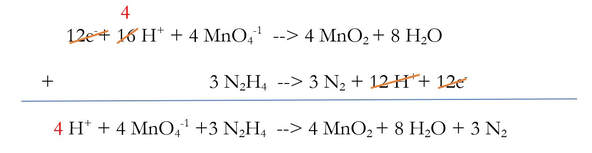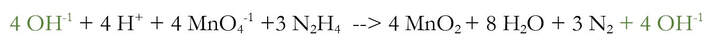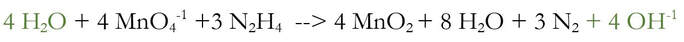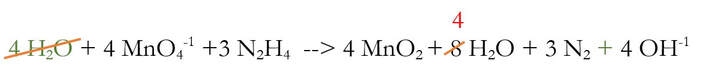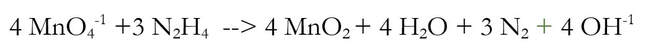Balancing Redox Reactions (part 3)
Yep, you guessed it. If some chemical reactions happen in an acidic environment, some others must happen in a basic one. So, we have to be able to balance basic redox as well. Fortunately, it is the same as acidic balancing, just with an added step on the end. So let's go through another example:
1) Divide the reaction into 2 half reactions, one that involves one element (Mn) and one that involves the other element (N):
2a) Balance all elements except H and O - they're already good, so we skip this step.
2b) Balance the oxygen by adding H2O (new step in blue)
2c) Balance the hydrogen by adding H+
3) Multiply each reaction by an integer to make the number of electrons equal.
Now, the new stuff. This reaction is balanced, but it's in an acidic solution, as evidenced by the extra H+ on the left. If the solution is basic, then there cannot be H+ in the solution, so we need to somehow make it basic. To make it basic:
6) Add enough OH-1 to the side of reaction to cancel out each of the H+. And, to keep the reaction balanced (because we spent a lot of time doing that), add the same amount of OH-1 to the other side of the reaction.
We added 4 OH-1 to each side of the reaction, because there were 4 H+ that needed to be removed. And when I say removed, I really mean neutralized. The 4 H+ and the 4 OH-1 will chemically react to make 4 H2O (water):