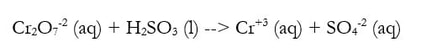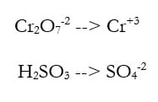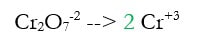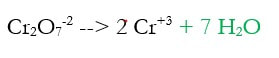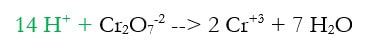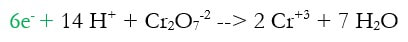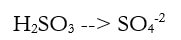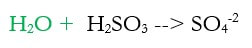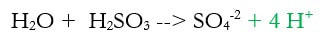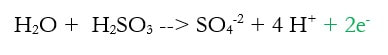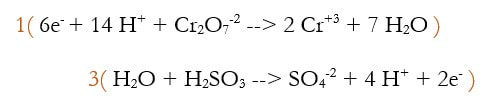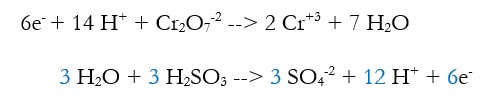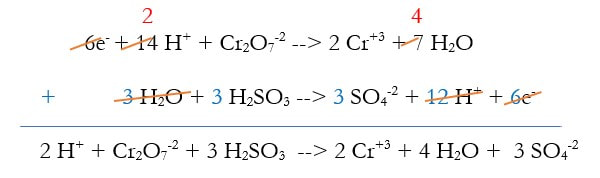Balancing Redox Reactions (part 2)
Acidic Redox Reactions
Some redox reactions have more than just single elements in them, or may have to occur in an acidic environment (in the presence of an acid). In these cases, we must take the balancing system we have previously learned and modify for the acidic conditions. For instance, say you need to balance this reaction:
Some redox reactions have more than just single elements in them, or may have to occur in an acidic environment (in the presence of an acid). In these cases, we must take the balancing system we have previously learned and modify for the acidic conditions. For instance, say you need to balance this reaction:
There are many issues, which we can solve by going through an altered version of what we did before:
1) Divide the reaction into 2 half reactions, one that involves one element (Cr) and one that involves the other element (S):
1) Divide the reaction into 2 half reactions, one that involves one element (Cr) and one that involves the other element (S):
Here's where it gets tricky. Let's do one half reaction at a time. For step 2, we have substeps.
2a) Balance all elements except H and O (new step in green)
2a) Balance all elements except H and O (new step in green)
2b) Balance the oxygen by adding H2O (new step in green)
2c) Balance the hydrogen by adding H+ (new step in green)
2d) Add electrons to make the charge equal on both sides (new step in green)
The charge on the left was +14 -2 = +12 and the charge on the right was 2 x +3 = +6. Electrons are negative, and can only bring charges down, so we need to add 6 electrons (-6) to the left to make both sides +6.
Now we need to repeat steps 2a - 2d for the other reaction:
2a) Balance all elements except H and O . In this case, the S is equal, so we jump to 2b.
Now we need to repeat steps 2a - 2d for the other reaction:
2a) Balance all elements except H and O . In this case, the S is equal, so we jump to 2b.
2c) Balance the hydrogen by adding H+ (new step in green)
2c) Balance the hydrogen by adding H+ (new step in green)
2d) Add electrons to make the charge equal on both sides (new step in green)
Now, steps 3 - 5 are the same as before:
3) Multiply each reaction by an integer to make the number of electrons equal.
3) Multiply each reaction by an integer to make the number of electrons equal.
4) Multiply the 2 reactions out:
5) Use Hess's Law and add the reactions together, eliminating anything that is on both sides. The whole purpose of this is to get rid of the electrons, and sometimes other things:
At this point, since there are so many places to make mistakes, it's good to check your charge and elements. I usually check the charge, and the hydrogens. If either of those are wrong, then it's all wrong. But if their both right, it's probably right. You could check them all, but that takes time.
In this case, the charge on the left is 2(+1) + -2 = 0, and the right is 2(+3) + 3(-2) = 0. The hydrogen are 2(1) + 3(2) = 8 on the left, and 2 (4) = 8 on the right.
In this case, the charge on the left is 2(+1) + -2 = 0, and the right is 2(+3) + 3(-2) = 0. The hydrogen are 2(1) + 3(2) = 8 on the left, and 2 (4) = 8 on the right.