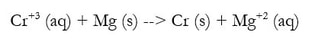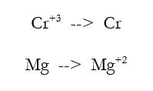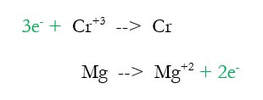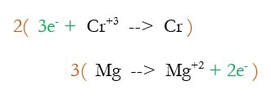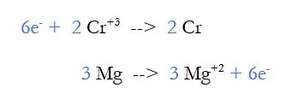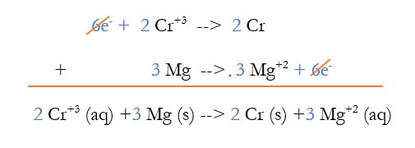Balancing Redox Reactions (part 1)
Redox reactions make up most of the chemical reactions we know: synthesis, decomposition, single replacement and combustion are all examples of redox reactions. In most cases, they are fairly easy to balance. But, when the reaction is written as a net ionic equation, it becomes a different animal. For example:
So the reaction already looks like it is balanced, but the charges are not. And in redox, the number of electrons being transferred is important. So we need to balance the charges as well. Now, you may have already figured out how to do it, but we need a process for all reactions, so here it is:
1) Divide the reaction into 2 half reactions, one that involves one element (Cr) and one that involves the other element (Mg):
1) Divide the reaction into 2 half reactions, one that involves one element (Cr) and one that involves the other element (Mg):
2) Balance each half reaction by adding the proper number of electrons to each side to make both sides of the reaction equal in charge. For instance, in the first reaction, charge on the left is +3, and the right side is neutral. You can add electrons to one side to bring the charge down. Since the left side of the reaction has to come down to make it equal to the right, we need to add 3 negative electrons to the left:
To the magnesium reaction, we added 2 electrons to the right to bring the charge of +2 down to 0. Helpful hint: for the two half reactions, you always add electrons to different sides in the two reactions (electrons go on one side for one reaction, and on the other side for the other).
3) Multiply each reaction by an integer to make the number of electrons equal. 2 and 3 both go into 6, so we multiply the first reaction by 2, and the second by 3:
3) Multiply each reaction by an integer to make the number of electrons equal. 2 and 3 both go into 6, so we multiply the first reaction by 2, and the second by 3:
4) Multiply the 2 reactions out:
5) Use Hess's Law and add the reactions together, eliminating anything that is on both sides. The whole purpose of this is to get rid of the electrons, and sometimes other things: