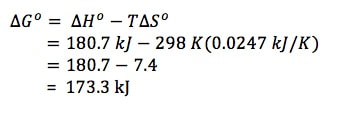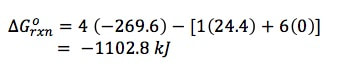Gibbs Free Energy
In our previous discussions, we have talked about how chemical reactions can release or gain heat (enthalpy) and how chemical reactions can become more disordered or more ordered (entropy). With these discoveries, scientists wanted to find a way to determine the spontaneity of a reaction. They knew it wasn't solely based on heat, because both endothermic and exothermic reactions were spontaneous. They also know it wasn't solely based on entropy, because disorder and order could both happen in spontaneous reactions. In fact, they both seemed to be af actor.
J. WIlliard Gibbs, a American mathematician, was the first to develop a process. Through algebraic manipulation of the Second Law of Thermodynamics, Gibbs was able to show that for a reaction
J. WIlliard Gibbs, a American mathematician, was the first to develop a process. Through algebraic manipulation of the Second Law of Thermodynamics, Gibbs was able to show that for a reaction
ΔG = ΔH - TΔS
where ΔG is called Gibbs free energy, or just free energy.
The free energy, ΔG, is a state function that essentially tells us whether a reaction is spontaneous or not. It is measured in kJ/mol. Using the equation, if you calculate the ΔG, then:
1) If ΔG is negative, the reaction is spontaneous in the forward direction.
2) If ΔG is zero, the reaction is at equilibrium.
3) If ΔG is positive, the reaction is spontaneous in the reverse direction (i.e., the forward reaction is not spontaneous).
Example 1: Calculate the standard free energy change for the reaction
The free energy, ΔG, is a state function that essentially tells us whether a reaction is spontaneous or not. It is measured in kJ/mol. Using the equation, if you calculate the ΔG, then:
1) If ΔG is negative, the reaction is spontaneous in the forward direction.
2) If ΔG is zero, the reaction is at equilibrium.
3) If ΔG is positive, the reaction is spontaneous in the reverse direction (i.e., the forward reaction is not spontaneous).
Example 1: Calculate the standard free energy change for the reaction
N2 (g) + O2 (g) --> 2 NO (g)
given that ΔHO = 181 kJ and ΔSO = 25 J/K. Is the reaction spontaneous or not?
Notice in the calculation above, we had to convert entropy to kJ/K in order for the units to align with enthalpy.
Standard Free Energy
Standard free energy is the free energy at 298 K. In the back of the text book and at this link, there is a list of standard thermodynamic data. Free energy is also listed there, which is the free energy of standard formation of those compounds. As with enthalpy and entropy, the standard free energy can also be calculated by:
Standard Free Energy
Standard free energy is the free energy at 298 K. In the back of the text book and at this link, there is a list of standard thermodynamic data. Free energy is also listed there, which is the free energy of standard formation of those compounds. As with enthalpy and entropy, the standard free energy can also be calculated by:
ΔGO = ∑ΔGOf (products) - ∑ΔGOf (reactants)
Example 2: Using data from Appendix 4, calculate the standard free energy change for the reaction
P4 (g) + 6 Cl2 (g) --> 4 PCl3 (g)
One AP question that almost always appears on the exam is one that is similar to this:
A certain chemical reaction is exothermic and increases its randomness. Is the reaction
a) Always spontaneous
b) Never spontaneous
c) Spontaneous only at high temperatures
d) Spontaneous only at low temperatures
This question is designed to see if you know the Gibbs free energy equation and whether you know how to use it.
|
For this question, you have to realize that since the reaction is exothermic, the ΔH is negative. Also, you must realize that the ΔS is positive because of the increase in randomness. When you put these into the ΔG = ΔH - TΔS equation, you find:
ΔG = ΔH - TΔS
ΔG =(-) - + (+) because the temperature is always positive on the Kelvin scale, you are taking a negative number and subtracting a positive number. No matter what the temperature is, the ΔG will always come out negative, making it a spontaneous reaction at any temperature. Since there are only 4 permutations of possibilities for signs of ΔH and ΔS, they are listed below: |