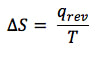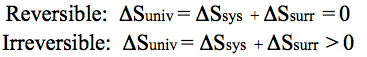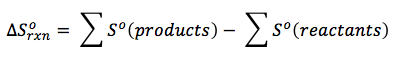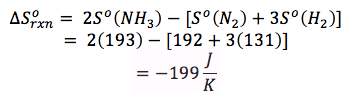Entropy
Scientists have a desire to be able to predict when a chemical reaction is spontaneous. These chemists believed originally that if a reaction released heat or did work, then the reaction would be spontaneous. However, there are some reactions that are spontaneous in which no work is done and there is no change in heat. So, scientists had to work on what might be the other factor that affects spontaneity. This led them to the concept of entropy.
Entropy is a measure of the disorder in a system. The symbol given to entropy change in a chemical reaction is ΔS. Entropy is a state function that is measured and calculated by the following equation:
We will never use this equation to calculate ΔS because we do not have the ability to perform this experiment in the lab. It is really hard to measure the heat given off from a reversible reaction when the temperature doesn't change. There are other ways.
Entropy measures disorder or chaos. This can be a confusing concept. Basically, when you knock over a jar of marbles, you are creating disorder. The marbles go everywhere. They were contained in an organized area and then they were released to go willy-nilly. This causes the entropy of the marble system to increase, as there is more disorder and chaos. An increase is entropy means a +ΔS. If you were to clean up the marbles again, you would be restoring the order, and decreasing the disorder, which is a -ΔS.
Entropy measures disorder or chaos. This can be a confusing concept. Basically, when you knock over a jar of marbles, you are creating disorder. The marbles go everywhere. They were contained in an organized area and then they were released to go willy-nilly. This causes the entropy of the marble system to increase, as there is more disorder and chaos. An increase is entropy means a +ΔS. If you were to clean up the marbles again, you would be restoring the order, and decreasing the disorder, which is a -ΔS.
The Second Law of Thermodynamics
The second law of thermodynamics has been used to explain why the universe is expanding. It essentially says that the entropy of the universe has to get bigger, as the universe is expanding. Therefore, in any reversible reaction, there is no overall change in entropy. So if the reaction decreases in entropy, then the surroundings have to increase in entropy by the same amount. If the reaction is irreversible, there has to be a overall increase in entropy. So if the reaction decreases in entropy, then the surroundings have to increase in entropy by an amount more than the entropy lost by the reaction. In mathematical terms, it is written:
The second law of thermodynamics has been used to explain why the universe is expanding. It essentially says that the entropy of the universe has to get bigger, as the universe is expanding. Therefore, in any reversible reaction, there is no overall change in entropy. So if the reaction decreases in entropy, then the surroundings have to increase in entropy by the same amount. If the reaction is irreversible, there has to be a overall increase in entropy. So if the reaction decreases in entropy, then the surroundings have to increase in entropy by an amount more than the entropy lost by the reaction. In mathematical terms, it is written:
All of this is difficult to understand. An example will make it easier. Suppose you try to clean the marbles up. As you clean the marbles up, you order the system, you decrease the entropy, or -ΔS. In order to do this job, you must move around, expend energy, burn food, etc. This causes entropy, or disorder, or chaos in the universe,+ΔS. This increase in entropy of the universe is more than the decrease in entropy you do to reorder the system and put the marbles back in the jar. The universe has an overall increase in entropy (has become more chaotic), even though a small part has decreased entropy (become more ordered).
Degrees of Freedom
Since entropy is randomness, a decrease in the randomness of a system means the system has become more ordered. To increase the order, the system must decrease the number of degrees of freedom. Degrees of freedom are the different ways objects can move. There are ways to change the degrees of freedom. They include
1) Forming or breaking bonds - There is no real pattern to this. More bonds sometimes means more entropy, and fewer bonds sometimes means more entropy. No matter what, changing the number of bonds will change the entropy.
2) Translational motion - This is the movement of the whole molecule. This generally increases with higher temperature.
3) Rotational motion - This is how the molecule spins as it moves. Again, it generally increases with higher temperature.
4) Vibrational motion - This is the movement of the bonds, either the lengthening or shrinking of the bond length, or the atoms moving closer and farther apart within the molecule. Again, it generally increases with higher temperature.
The Third Law of Thermodynamics
The main way to decrease the degrees of freedom (and the entropy) of a system is to decrease the temperature. As you cool the substance, the rotational, translational and vibrational motion all slow. If we keep lowering the temperature, the entropy decreases. Theoretically, at some point, the temperature will get so cold that there is no entropy - no chaos. This occurs at absolute zero. This is the third law of thermodynamics. When all motion stops, there will be no entropy. This only occurs at absolute zero, or 0 Kelvin. No entropy means a perfect crystalline structure in the solid and there would be no vibrations at all. Which means no entropy.
Entropy Changes in Chemical Reactions
Since entropy is randomness, a decrease in the randomness of a system means the system has become more ordered. To increase the order, the system must decrease the number of degrees of freedom. Degrees of freedom are the different ways objects can move. There are ways to change the degrees of freedom. They include
1) Forming or breaking bonds - There is no real pattern to this. More bonds sometimes means more entropy, and fewer bonds sometimes means more entropy. No matter what, changing the number of bonds will change the entropy.
2) Translational motion - This is the movement of the whole molecule. This generally increases with higher temperature.
3) Rotational motion - This is how the molecule spins as it moves. Again, it generally increases with higher temperature.
4) Vibrational motion - This is the movement of the bonds, either the lengthening or shrinking of the bond length, or the atoms moving closer and farther apart within the molecule. Again, it generally increases with higher temperature.
The Third Law of Thermodynamics
The main way to decrease the degrees of freedom (and the entropy) of a system is to decrease the temperature. As you cool the substance, the rotational, translational and vibrational motion all slow. If we keep lowering the temperature, the entropy decreases. Theoretically, at some point, the temperature will get so cold that there is no entropy - no chaos. This occurs at absolute zero. This is the third law of thermodynamics. When all motion stops, there will be no entropy. This only occurs at absolute zero, or 0 Kelvin. No entropy means a perfect crystalline structure in the solid and there would be no vibrations at all. Which means no entropy.
Entropy Changes in Chemical Reactions
One more thing we want to be able to do with entropy is to be able to somewhat predict the entropy changes of a chemical reaction. First of we must remember that nothing has an entropy of zero, unless it is at absolute zero. But, a chemical reaction can have a -ΔS. You can predict the molar entropies of a substance based on the following trends:
1) More molecules on a side of the reaction will mean that that side of the reaction will have more entropy.<br>
2) Gases have more entropy than liquids which have more entropy than solids.
3) Larger molar masses mean more entropy.
4) More atoms in a formula means more entropy.
Let's try a few reactions and predict the entropy changes:
2) Gases have more entropy than liquids which have more entropy than solids.
3) Larger molar masses mean more entropy.
4) More atoms in a formula means more entropy.
Let's try a few reactions and predict the entropy changes:
|
1) N2 (g) + 3 H2 (g) --> 2 NH3 (g)
2) CaCO3 (s) --> CaO (s) + CO2 (g) 3) NH3 (g) + HCl (g) --> NH4Cl (s) |
1) In this case, we go from 4 moles of reactant to 2 moles of product. Because there are less moles of materials, it is more organized, which reduces chaos, making entropy decrease.
2) There are more moles of product and, more importantly, the reaction makes gas (which is more chaotic) from solid. The entropy is increasing. 3) Decrease in entropy - fewer molecules and it is a solid, both of which cause more order and less chaos. |
Standard Entropy in Reactions
Scientists have determined the amount of entropy in many common substances used in chemical reactions at standard conditions. These entropies are listed in the back of most textbooks. If a reaction happens at standard temperature, 298 K, you can determine the entropy change of the chemical reaction by subtracting the standard entropy of all the products minus that of the reactants, or
Scientists have determined the amount of entropy in many common substances used in chemical reactions at standard conditions. These entropies are listed in the back of most textbooks. If a reaction happens at standard temperature, 298 K, you can determine the entropy change of the chemical reaction by subtracting the standard entropy of all the products minus that of the reactants, or
Example: Calculate the change in the standard entropy, ΔSO for the synthesis of NH3 from its elements at 298 K.
N2 (g) + 3 H2 (g) --> 2 NH3 (g)
|
To solve, we must first find the standard entropies for all the components of the reaction from the back of the book. Then, we also have to multiply the entropies by the moles of each in the balanced reaction.
N2: ΔSO = 192 J/mol K
H2: ΔSO = 131 J/mol K NH3: ΔSO = 193 J/mol K |
The fact that the entropy is negative proves our prediction from above.
|
One other thing to notice: All of the entropies of the materials in the reaction are positive and not zero. Aswe discussed earlier, entropies for substances cannot be negative, and they can only be zero at absolute zero. A -ΔS can be possible for reactions or changes, because a reaction can become more ordered. But, a substance cannot have a -ΔS or ΔS=0.