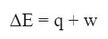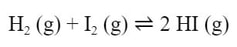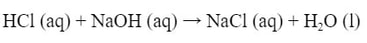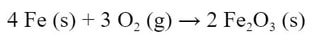Spontaneous and Reversible Reactions
Spontaneous, or Thermodynamically Favored
The first law of thermodynamics is what we commonly know as the law of conservation of energy. It says that the amount of energy in the universe is a constant number, so energy cannot be created or destroyed. However, it can be transformed from one kind of energy into another. Or energy can be moved from one place to another. In other words, there can be a change in energy of one object, but the overall energy is constant as the energy is transferred. Mathematically, the first law is written like this:
The first law of thermodynamics is what we commonly know as the law of conservation of energy. It says that the amount of energy in the universe is a constant number, so energy cannot be created or destroyed. However, it can be transformed from one kind of energy into another. Or energy can be moved from one place to another. In other words, there can be a change in energy of one object, but the overall energy is constant as the energy is transferred. Mathematically, the first law is written like this:
In the grand scheme of things, this basically means that if there is a change in energy of something (ΔE) that either some heat (q) must be generated or work (w) has to be done, or a combination of the two. This equation does not have a lot of useful mathematical applications, it is more of a concept.
Despite this law, there are many chemical reactions that happen despite energy being conserved in the reaction. These reactions are said to be spontaneous, or thermodynamically favorable. Reactions that happen of their own accord when the chemicals are combined are said to spontaneous. Reactions that do not occur, or chemicals added together that do not react, are called not spontaneous, or not thermodynamically favorable. This concept is shown best by using examples:
Iron rusting is thermodynamically favorable. It will inevitably happen. Rusted iron "unrusting" is not thermodynamically favorable.
Water freezing is spontaneous below zero degrees Celsius. Water melting is spontaneous above zero degrees Celsius.
Water freezing is spontaneous below zero degrees Celsius. Water melting is spontaneous above zero degrees Celsius.
Reversible Reactions
The idea of reversible reactions is not the same as spontaneous and not spontaneous. It is important to know the distinction. A reversible process is one that happens by doing something, and the by doing the exact opposite the process will reverse. For example, you can freeze water by removing enough heat. If you add that heat back in (exact opposite of removing heat), the process will reverse and the water will melt.
Reactions in equilibrium are reversible. In the reaction:
The idea of reversible reactions is not the same as spontaneous and not spontaneous. It is important to know the distinction. A reversible process is one that happens by doing something, and the by doing the exact opposite the process will reverse. For example, you can freeze water by removing enough heat. If you add that heat back in (exact opposite of removing heat), the process will reverse and the water will melt.
Reactions in equilibrium are reversible. In the reaction:
If you add hydrogen to iodine, the reaction will form hydrogen iodide. If you remove the hydrogen, the reaction will reverse and the hydrogen iodide will decompose back into hydrogen and iodide, according to Le Chatelier.
Some reactions are irreversible. When you do this reaction:
Some reactions are irreversible. When you do this reaction:
The HCl and NaOH will react to form salt and water. This reaction will not reverse itself. If you separate HCl from NaOH, it will not reverse. Nor does adding NaCl and water together make HCl and NaOH. The reaction cannot be reversed. It is considered irreversible.
That doesn't mean you cannot reclaim the reactants of a irreversible reaction. You can, you just have to go through a different process. Iron will rust. It is spontaneous and irreversible. If you want to change rust back into iron, it can be done, just not in the exact opposite way. The rusting of iron happens according to this reaction.
That doesn't mean you cannot reclaim the reactants of a irreversible reaction. You can, you just have to go through a different process. Iron will rust. It is spontaneous and irreversible. If you want to change rust back into iron, it can be done, just not in the exact opposite way. The rusting of iron happens according to this reaction.
If you add oxygen to iron, it will form rust (iron (III) oxide). If you place rusted iron in an environment where there is no oxygen, it will not "unrust". But, there are other chemical reactions you can do to restore the iron. Irreversible reactions can be reversed, just in a different process than how the reaction was done in the first place.