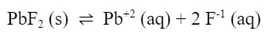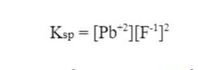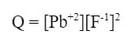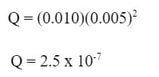Ion Product
Sometimes, for these slightly soluble solids, we wonder how many of each ion we need to add to make a precipitate. For instance, if we are combining Pb+2 ions and F-1 ions from 2 different solutions, depending on how much of each ion we use, we may or may not form a precipitate. If there is not enough of one of the ions, then a precipitate won't form. But if we add enough, it will form. So how are we going to know if the ions we added are enough to precipitate?
This is the situation where we calculate an ion product. Ion product is what it says: Multiply the number of ions in the solution according to the Ksp. If the ion product, Q, is different from the Ksp, then we can determine whether a precipitate will form. Let's do an example:
Example: If a 0.01 M Pb(NO3)2 solution is mixed with a 0.005 M NaF solution, will a precipitate of PbF2 form? (Ksp for PbF2 is 3.6 x 10-8)
|
First, we need to review the Ksp expression for lead (II) fluoride:
Since, we are calculating the ion product for lead (II) fluoride, we simply take out the Ksp and replace it with Q.
So, we are given that the Pb(NO3)2 solution is 0.01 M, that means the Pb+2 concentration is 0.01 M. Also, we are told the NaF solution is 0.005 M, so F-1 concentration is 0.005 M.
We can put these concentrations in for the [Pb+2] and [F-1] in the ion product to get: When we solve for Q, we get:
In this case, the Q we calculate (2.5 x 10-7) is a bigger number than the Ksp (3.6 x 10-8), this means the product of ions are more than the maximum solubility of the ions for PbF2, so a precipitate will form.
|
There are 3 possibilities for Q.
- If Q = Ksp, then the system is at equilibrium, and there are the maximum number of ions to be soluble and a saturated solution with no precipitate is made
- If Q > Ksp, there are too many ions in solution for soluble equilibrium, so the extra ions will precipitate.
- If Q < Ksp, there are less ions in solution than needed to establish equilibrium, or not enough ions, so there is no precipitate.