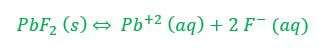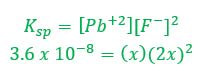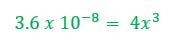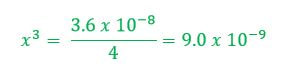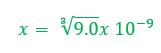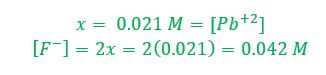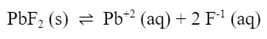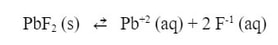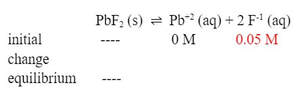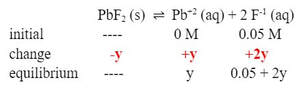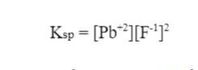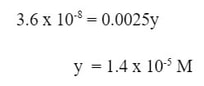Applications of Solubility Product
Determining Molar Solubilities
The other type of problem we need to be able to do is determine the molarity of the ions when given the solubility product.
Example 1: What concentration of ions will be present in a saturated solution of PbF2 (Ksp = 3.6 x 10-8)
Example 1: What concentration of ions will be present in a saturated solution of PbF2 (Ksp = 3.6 x 10-8)
Another example:
Molar Solubilities and the Common Ion Effect
When a slightly soluble substance is put in a solution where a common ion is present, the solubility of the material goes down. This is because the common ion in the solution pushes the equilibrium back toward the solid. Let's use an example. Suppose you tried to dissolve the lead (II) fluoride in water, according to example 1. We have already determined that the concentration of ions in solution.
When a slightly soluble substance is put in a solution where a common ion is present, the solubility of the material goes down. This is because the common ion in the solution pushes the equilibrium back toward the solid. Let's use an example. Suppose you tried to dissolve the lead (II) fluoride in water, according to example 1. We have already determined that the concentration of ions in solution.
But what if we tried to dissolve the same lead (II) fluoride in a solution of sodium fluoride. The fluoride ion in the solution would push the equilibrium to the left:
Which means that less lead (II) fluoride would dissolve. The (-x) for lead fluoride would be less. Let's do an example to show the effect:
Example 2: What is the molar solubility in a saturated solution of PbF2 (Ksp = 3.6 x 10-8) when placed in a solution of 0.05 M NaF?
|
Again, this is another ICE problem, but for this one, the initial concentration of F-1 ion is no longer zero, but 0.05 M F-1:
Now, the change is different, so we will use y as our change:
Whatever the change in F-1 is, it is going to be much smaller than the 0.05 M of the F-1, so we can remove the +y from the F-1:
Then, we solve for y, which will be the concentration of [Pb+2], and the molar solubility of PbF2:
|