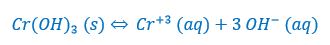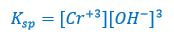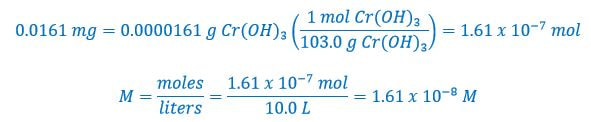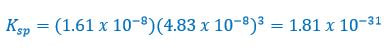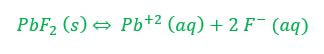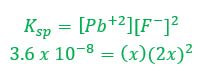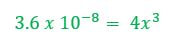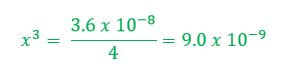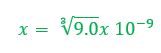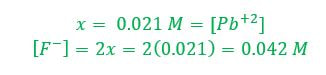Solubility Product
Dissolving and Precipitation
When an ionic compounds dissolves, it breaks up into ions. An example of this process can be shown with an equation like this:
When an ionic compounds dissolves, it breaks up into ions. An example of this process can be shown with an equation like this:
K2CrO4 (s) --> 2 K+ (aq) + CrO4-2 (aq)
Notice that in this reaction, as the substance dissolves, it produces 2 ions of potassium because of the number of potassium ions in the original compound. This process is called dissolving. When this process involves ions, it is also called dissociation, as the solid not only becomes aqueous, it also breaks up into ions. If it didn't break up into ions (like molecular compounds don't), then it is simple dissolving.
When the reverse process occurs, it is called precipitation. In precipitation, two aqueous ions are introduced to one another and the ions come together to form a solid. An example would be when you mix aqueous lead nitrate with aqueous sodium chloride. The net ionic reaction for the precipitation is:
When the reverse process occurs, it is called precipitation. In precipitation, two aqueous ions are introduced to one another and the ions come together to form a solid. An example would be when you mix aqueous lead nitrate with aqueous sodium chloride. The net ionic reaction for the precipitation is:
Pb+2 (aq) + 2 Cl- (aq) --> PbCl2 (s)
As you can see, these are opposite processes. There are sometimes situations in which these two processes happen simultaneously and establish an equilibrium. Think of it this way: At 298 K, the maximum amount of salt that can dissolve in 100 g of water is 35 grams. After you put in 35 grams, no more salt will dissolve. If you put 40 g of salt in the water, the remaining 5 g of salt would not dissolve, right? Not quite.
What actually happens is the salt continues to dissolve. But, how is this possible? You can see the salt is not dissolving - it stays at the bottom of the beaker. The same amount is always there. However, kinetic molecular theory says that the salt continues to dissolve as the salt is ionized by the water. However, the water can only hold so much salt, so at the same time the water is dissolving the salt, some other salt is precipitating onto the surface of the salt. The same amount dissolves as precipitates. This sets up an equilibrium that could be written like this:
What actually happens is the salt continues to dissolve. But, how is this possible? You can see the salt is not dissolving - it stays at the bottom of the beaker. The same amount is always there. However, kinetic molecular theory says that the salt continues to dissolve as the salt is ionized by the water. However, the water can only hold so much salt, so at the same time the water is dissolving the salt, some other salt is precipitating onto the surface of the salt. The same amount dissolves as precipitates. This sets up an equilibrium that could be written like this:
NaCl (s) <==> Na+ (aq) + Cl- (aq)
Since this is an equilibrium, we can then create an equilibrium constant off of the equation. The K for this equation would be:
Ksp = [Na+][Cl-]
Notice that NaCl is not in the equation because it is a solid. Also, we label this as Ksp because we call this the solubility product. The Ksp is called the solubility product because we are multiplying (product) the concentrations of ions from the substance that dissolved.
For a substance like salt, the Ksp is going to be very large because salt is very soluble. We usually do not use Ksp for something that dissolves a lot. For highly soluble substances, a mass percentage makes more sense. We typically use Ksp for solids that are only slightly soluble in water, and have a low Ksp. Substances like Fe(OH)2 and Ca3(PO4)2.
For a substance like salt, the Ksp is going to be very large because salt is very soluble. We usually do not use Ksp for something that dissolves a lot. For highly soluble substances, a mass percentage makes more sense. We typically use Ksp for solids that are only slightly soluble in water, and have a low Ksp. Substances like Fe(OH)2 and Ca3(PO4)2.
Calculating the Solubility Product
There are essentially four kinds of calculations that we need to be able to do with this idea of solubility product. We are going to deal with two of them here, and the other two on the net page. The first is determining the solubility product from knowing the amount of material that dissolved.
Chromium (III) hydroxide is a very insoluble salt. In 10.0 L of water, only 0.0161 mg of the solid will dissolve. What is the solubility product for chromium (III) hydroxide?
So, first we write the dissociation equation for solid chromium (III) hydroxide:
So, first we write the dissociation equation for solid chromium (III) hydroxide:
And then we write a solubility product expression, Ksp:
Next, we need to determine the molarity of each ion. To do this we need to determine the moles from the mass of chromium (III) hydroxide:
Now, 1.61 x 10-8 M is the concentration of Cr(OH)3 that dissolved, but not the ions. The ions of [Cr+3] will be 1.61 x 10-8 M, because the mole ratio of the Cr(OH)3 to Cr+3 is 1 to 1 from the chemical reaction above. But, the [OH-] concentration is 3 times as much, because there are 3 ions of OH- in the original compound. Therefore, the [OH-] is 3 x 1.61 x 10-8 = 4.83 x 10-8 M.
Now, if we plug in the M of the ions in the Ksp expression:
Now, if we plug in the M of the ions in the Ksp expression:
Another Example:
The other type of problem we need to be able to do is determine the molarity of the ions when given the solubility product.
Example 2: What concentration of ions will be present in a saturated solution of PbF2 (Ksp = 3.6 x 10-8)
Example 2: What concentration of ions will be present in a saturated solution of PbF2 (Ksp = 3.6 x 10-8)
|
Then, we must fill in the ICE table. Since PbF2 is a solid, we need not worry about the amount of it, since it won't figure into the equilibrium calculation. Pb+2 and F- both start at zero before the solid dissolves.
When an unknown amount of PbF2 dissolves, the amount is going to go down by x. Therefore the change in PbF2 is -x. Pb+2 will go up by x and F- will go up by 2x (based on the balanced chemical reaction). The amount of [Pb+2] = x and [F-] = 2x. |
Another example: