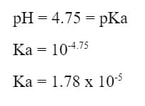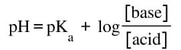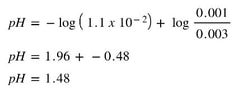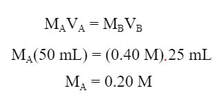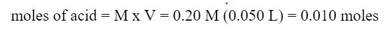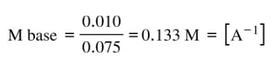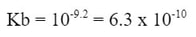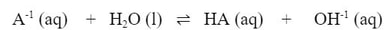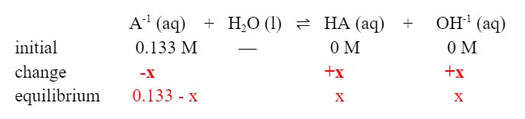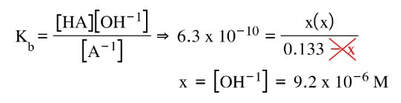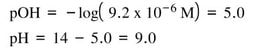Weak Acid Titrations
Titration Curves for Weak Acids
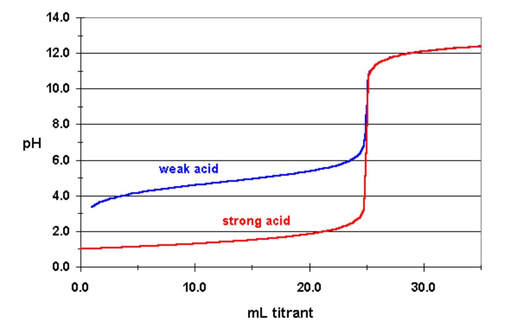
Titration curves for weak acids look different than strong acids. Below is a comparison between a strong acid and weak acid curve on the same graph:
Titration curves for weak acids look different than strong acids. Below is a comparison between a strong acid and weak acid curve on the same graph:
There are 4 key differences that need to be recognized between a weak acid curve:
- The weak acid will start at a higher pH (with no titrant added). This is because it is a weak acid and doesn't dissociate completely and has a lower concentration of hydrogen ion in solution.
- The weak acid will have a more gradual increase in pH before equivalence. This is because as the acid reacts with the added base, there is a formation of the conjugate base. The conjugate base will make the solution a buffer and increase the pH a little more as we get closer to the equivalence point.
- The jump in pH at the equivalence point will be smaller. Since the pH starts higher for a weak acid, and because you are making the cojugate base, the pH just before the jump will be higher, therefore has a smaller jump to get to the base's pH.
- And, most importantly, the pH at the equivalence point will NOT be 7. It will be above 7. This is because although the base solution has neutralized all the acid at the equivalence point, and there is no acid, there is conjugate base. In fact, the solution ONLY has conjugate base at equivalence. Because of the conjugate base, it will raise the pH above 7. You can use the Kb of the base to determine the pH at equivalence. You just need to figure out the molarity of the weak base from the neutralization. (More on that later).
|
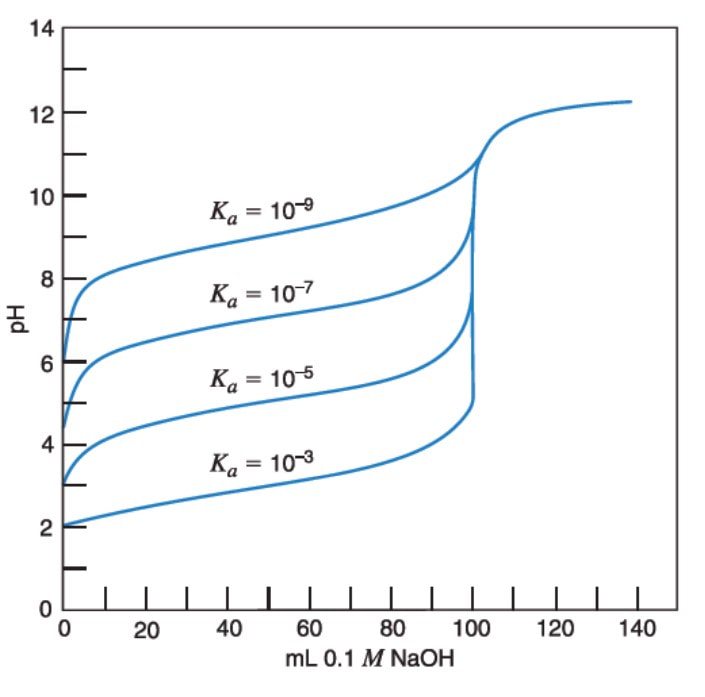
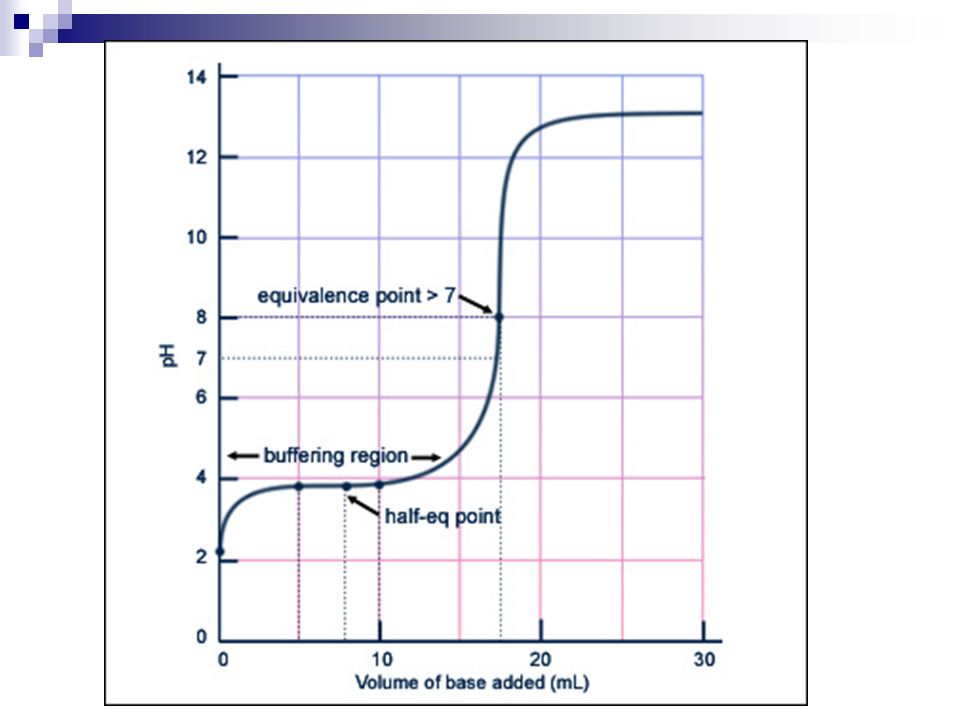
In addition to these differences, the weaker the acid, the farther from a strong base titration curve:
Notice in the image to the right, the weaker the smaller the Ka, the higher the pH starts, the more gradual the pH change before equivalence and the smaller the jump at equivalence. Also, as you can see from this diagram, the weaker the acid, the higher the pH will be at equivalence. From a titration curve graph, you can estimate the pH at equivalence. In order to do this, you simply estimate the pH halfway through the jump. For the Ka = 10-5, the pH starts to jump at about 7 and finishes its jump about 10.6. Midway though that jump would be at 8.8.
Again, this is just an estimate. In order to find the true pH, you would need the molarity of the conjugate base you are making and the Kb of the base |
Other Pieces of Information Gleaned from a Weak Acid Titration Curve
|
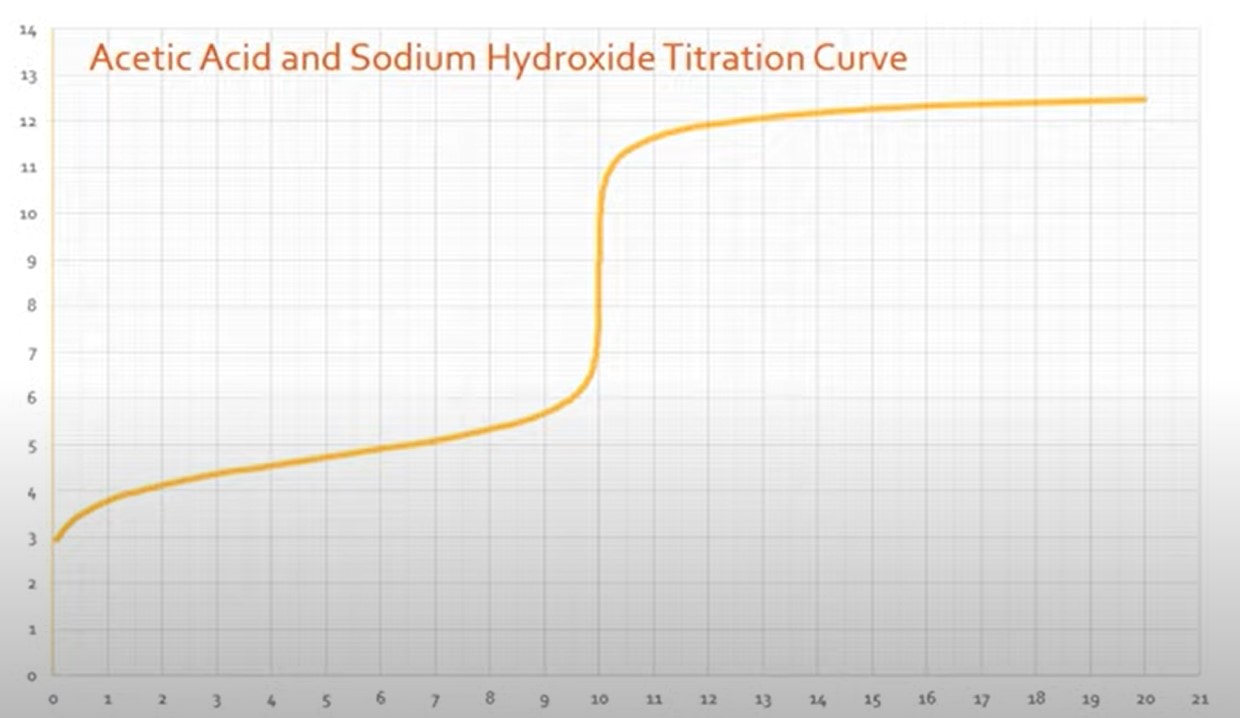
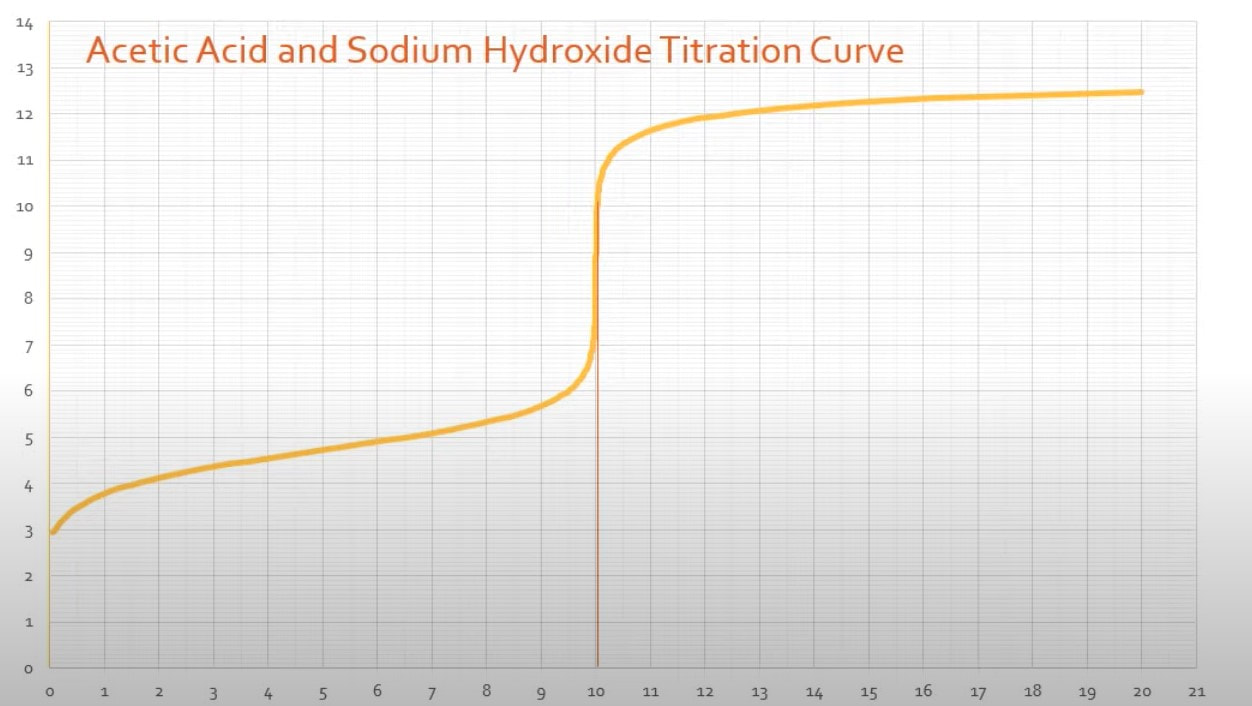
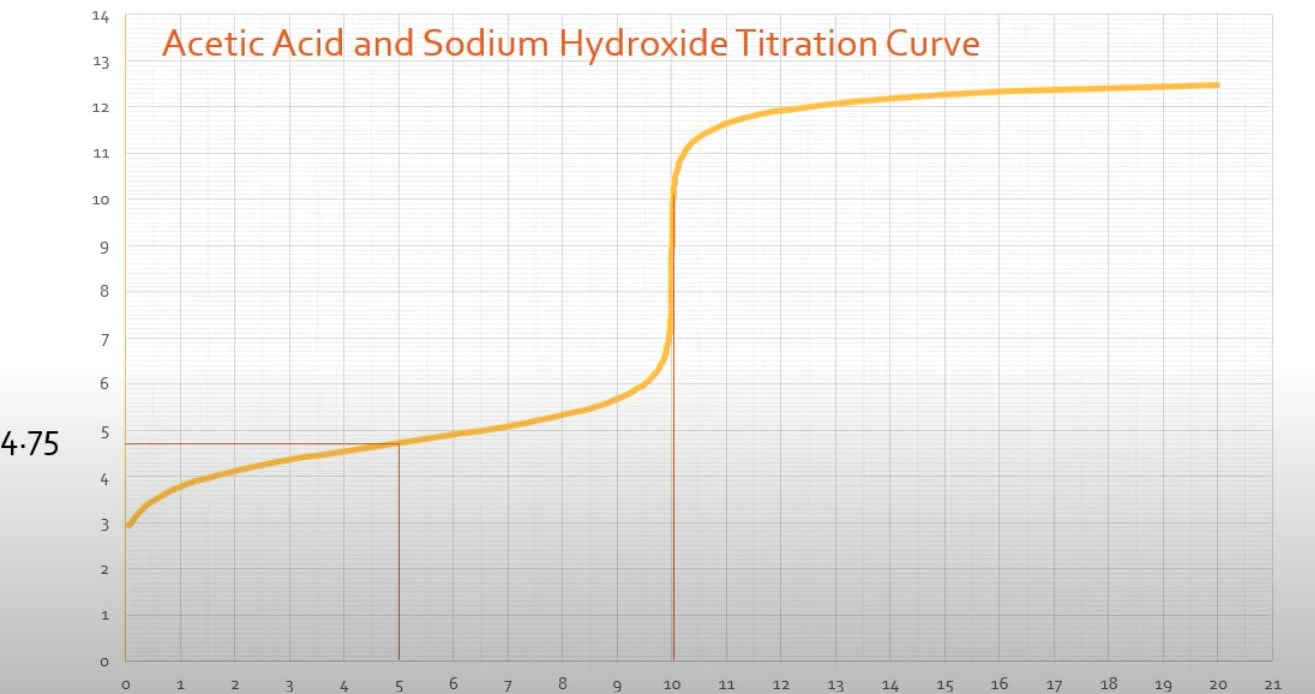
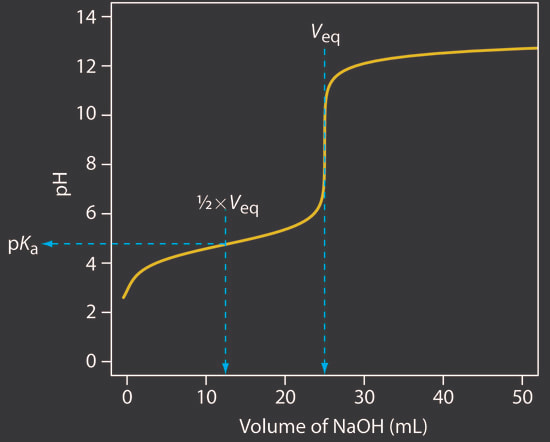
There are a couple other things that you can identify about an acid from its titration curve: 1) Ka - The Ka of an acid can be determined from the titration curve. When you have a titration curve like the one to the right (Fig A): When you look at the graph, you can determine the equivalence point happens at the jump, which is at 10 mL of base added. (Fig B) Since the equivalence point occurs at 10 mL, the half-equivalence point happens at 5 mL. (Fig C) At 5 mL, the pH of the solution is 4.75, according to the graph. At that point, the solution is exactly half neutralized, which means that there are equal amounts of weak acid (leftover that hasn't been neutralized) and weak conjugate base (made from the neutralization of the acid). At this point, the half equivalence point, the measured pH = pKa. So, since the pH is 4.75, we can determine the Ka of the acid: |
Fig A
Fig B
Fig C
|
|
2) Buffers
On a titration curve, the region before equivalence is considered the buffer region. Meaning, once you start adding base and up until the equivalence point, there is a combination of acid and conjugate base. This means that the pH in this region is not only caused by the mixture of acid and conjugate base, the pH at any point on that curve can be calculated using the buffer equation: The nice part about finding the pH in the buffer region is
a) you can use the equation and b) the numbers in the base and acid can just be in moles because the volumes are together, so they will cancel. |
Example: Suppose 30 mL of a 0.10 M HNO2 solution (Ka = 1.1 x 10-2) is being titrated with 0.20 M KOH. What is the pH when 5.0 mL of KOH has been added?
Well, if we calculate the moles of acid and base that have been added:
Then, we use the Ka and the moles, we can determine the pH using the equation:
Now, based on the graph, it looks like the pH at equivalence is going to be about 9. However, we can get a more accurate number. The pH at the equivalence point is going to be solely dependent on the conjugate base of the acid. Which means we need to find the molarity of that conjugate base and use the Kb of the base to determine the exact pH.
a) To determine the molarity of the conjugate base, we know the moles of conjugate base is going to have to be equal to the moles of acid used, so:
a) To determine the molarity of the conjugate base, we know the moles of conjugate base is going to have to be equal to the moles of acid used, so:
To get to molarity, we divide by the total volume of 75 mL, or 0.075 L:
The pKa for the acid is about 4.8. So the pKb for the base is 14 - 4.8 = 9.2. If we undo the pKb, the Kb would be:
Now, we don't really know what the acid or base is, so we can use the generic A-1, and write the equation:
If we put in an ICE table below the equation, we can then fill it in:
And then plug in the values into the Kb expression and solve for [OH-1]:
Then solve for pOH and pH: