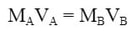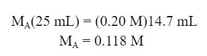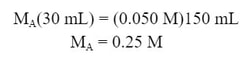Titrations
Titrations are a process to identify the unknown concentration of a solution. In a titration, you slowly add a known concentration of a titrant (something that reacts with what you are testing) to an unknown concentration solution. Then, through the use of stoichiometry, you would use the data to determine the unknown concentration of the solution. You would typically use a buret to add the titrant, that way you can know exactly how much titrant you use for reaction.
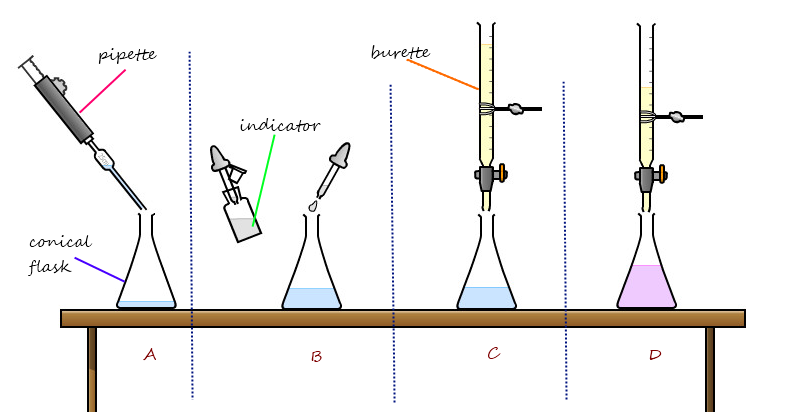
Titrations can be done in any type of solution reaction, you just need some sort of indicator that tells when the reaction is complete. Titrations are typically used to determine the unknown concentration of an acid or a base by using an indicator to tell the equivalence point of the titration. For example, you can find the concentration by following the steps in the diagram below:
Titrations can be done in any type of solution reaction, you just need some sort of indicator that tells when the reaction is complete. Titrations are typically used to determine the unknown concentration of an acid or a base by using an indicator to tell the equivalence point of the titration. For example, you can find the concentration by following the steps in the diagram below:
- a) Using a pipet, measure out a small amount of unknown concentration acid to be tested. Make sure you know what volume of acid you used.
- b) Add an indicator to the acid solution so that you can tell when you have reached equivalence. When titrating to find the concentration of an acid, phenolphthalein is a good choice.
- c) Fill a buret with known concentration base. Slowly drain the base into the acid. As the base becomes more prominent, a color change should occur. When the color starts to change, swirl the flask. If the color goes away, then acid is more prominent than base. Keep adding base.
- d) When the color change is faint but constant (when you swirl the color doesn't go away), you have reached equivalence. Record the amount of base needed to react all the acid.
From this data, you can determine the molarity of the acid. Let's use an example to demonstrate how the data is used:
Example #1: 25 mL of an unknown concentration HCl solution is titrated. 0.20 M NaOH is used as a titrant. The phenolphthalein indicator turns a light pink color when 14.7 mL of NaOH has been added. What is the molarity of the acid?
First, we need to know the mole ratio. If the mole ratio of acid to base is 1:1 (which we will typically use), the equation you can use is:
In this equation, the moles of acid and base are equal, which is true at the equivalence point. So, by plugging in the data we know, we can solve for Ma:
Titration Curves
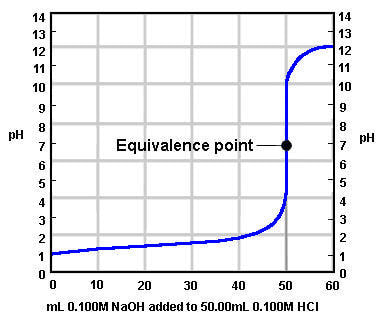
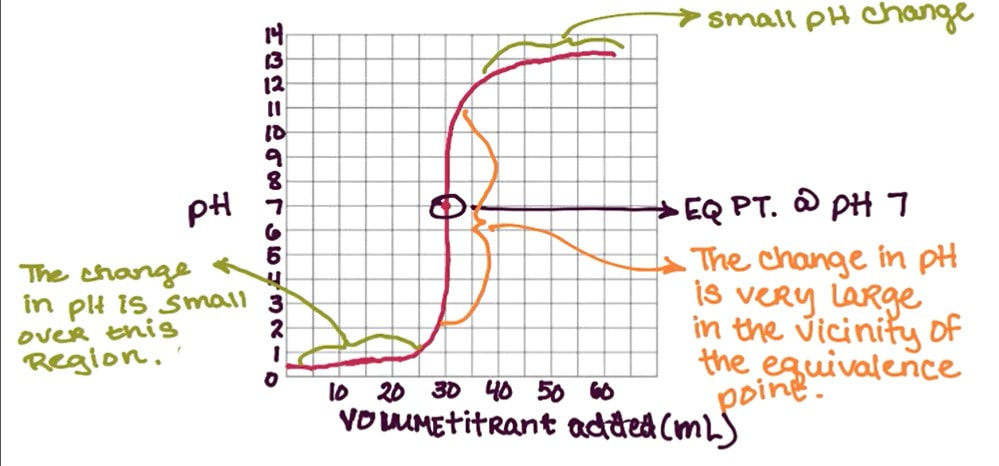
A titration curve is a plot of pH (y-axis) versus titrant added to achieve neutralization. A typical strong acid titration curve in which a strong base is added to it looks like this:
A titration curve is a plot of pH (y-axis) versus titrant added to achieve neutralization. A typical strong acid titration curve in which a strong base is added to it looks like this:
Some things to notice about the graph:
It is possible to use the titration curve to determine the molarity of the unknown acid. The jump in pH corresponds to the equivalence point. If you read the x-axis (titrant added) at the jump, you can identify the volume of base needed to reach equivalence. That coupled with the volume of acid used and molarity of the base will allow you to determine the unknown concentration like the example above.
- The pH of the acid starts low, before any base is added.
- The pH of the mixture before equivalence stays low because as the acid is neutralized by the base, the base is the limiting reactant. Once the base is reacted, the leftover acid controls the pH.
- There is a large jump in pH. The large jump occurs at the equivalence point, where the moles of acid = moles of base. At this point, you have added the appropriate amount of base to neutralize all the acid, and the pH at equivalence is 7.
- After the equivalence point, the pH is high because now the acid is the limiting reactant. The excess base controls the pH.
- All titration curves for strong acid/strong bases look the same. The small differences in the curve are
- where the jump in pH occurs depends on the relationship between the molarity of the acid and the base.
- what the starting pH is depends on the initial concentration of the acid
It is possible to use the titration curve to determine the molarity of the unknown acid. The jump in pH corresponds to the equivalence point. If you read the x-axis (titrant added) at the jump, you can identify the volume of base needed to reach equivalence. That coupled with the volume of acid used and molarity of the base will allow you to determine the unknown concentration like the example above.
|
Here's another example:
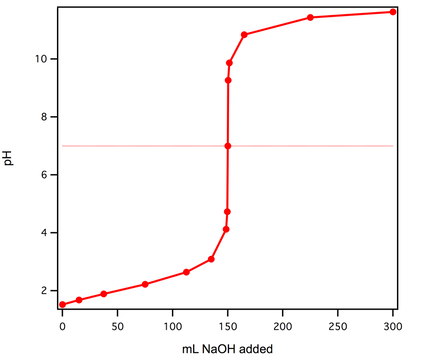
Example #2: A 30 mL sample of unknown concentration acid has the following titration curve, when titrated with 0.050 M NaOH. Determine the molarity of the unknown acid. In the titration to the right, the pH jumps at 150 mL. The concentration of the base is 0.050 M. Using that data and the 30 mL acid: |