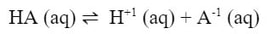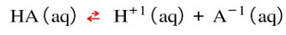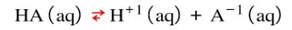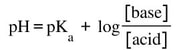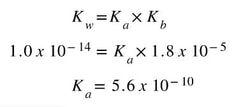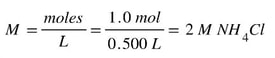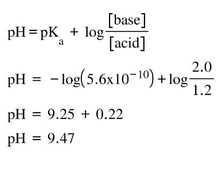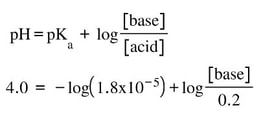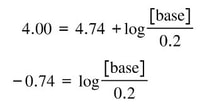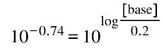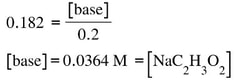Buffers
Buffers are solutions that resist changes in pH when acids or bases are added. The use of a buffer is necessary when you want the pH to stay the same if a small amount of acid or base are added to it. This kind of solution can be made by mixing an acid with its conjugate base. By doing this, the acid and conjugate base establish an equilibrium between them. In the example below:
You would make the solution by starting with a solution of HA. Then, you would add the conjugate base, like NaA (which contains the A-1 ion). The two solutions would establish an equilibrium. Then, if a small amount of acid were added (H+1 ions) into the solution, the equilibrium would consume the added H+1 ions and reverse the reaction. According to Le Chatelier, the reaction would reverse and the products would go down to reestablish equilibrium:
Well, this would decrease the amount of H+1 ions, which would keep the pH about the same. The pH would not change because the added H+1 ions would be converted to HA, keeping the H+1 ions about the same, maintaining the pH.
Likewise, if a small amount to base were added, the OH-1 ions would react with the H+1 ions to form water. This would decrease the H+1 ions in the solution, and more HA would dissociate to keep the pH about the same:
Likewise, if a small amount to base were added, the OH-1 ions would react with the H+1 ions to form water. This would decrease the H+1 ions in the solution, and more HA would dissociate to keep the pH about the same:
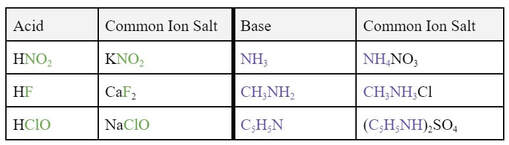
In order for the buffer to work properly, you need an acid with its conjugate base, or a base with its conjugate acid. The conjugates would need to be made from a salt that contains the conjugate. A buffer is a direct application of the common ion effect, and some examples of acid/salt or base/salts are listed here:
.Properties of Buffers
We've discussed how you make buffers. Properties of buffers include
We've discussed how you make buffers. Properties of buffers include
- Buffers can have very different pHs. For instance, if you make a buffer using an acid that has a higher Ka than its conjugate base's Kb, the buffer will be acidic. By contrast, a buffer made with a base that is stronger than its conjugate acid will be basic.
- Buffers made with bases that are stronger than their conjugate acids will be better at buffering acids. Buffers made with acids that are stronger than their conjugate bases will be better at buffering bases.
- The more moles of acid/conjugate base in the solution, the greater the buffer capacity. Buffer capacity is a measure of how much acid or base you can add to the buffer solution before it changes pH significantly (usually by 1 unit of pH). A 1M acid/1 M conjugate base mixture will be able to buffer longer than a 0.1 M acid/0.1 M base solution. Another way of looking at this is the more acid and conjugate base that are in solution, the more acid or base you can add before the pH changes.
- Buffer pH is dependent on the Ka/Kb of the primary acid/base, and the comparative concentrations of the two. If you have equal amounts of acid and base, the pKa = pH of the buffer. If you have more base, the pH will be higher. If you have more acid, the pH will be lower. In most cases, depending on how much acid and base you add, you can make whatever pH you want. As long as you use an acid or base close to the desired pH. The pH of a buffer can be calculated using the following equation:
The pH of a buffer can be easily determined using this equation. Let's look at an example:
a) A 500 mL solution of ammonia is made so that it has a concentration of 1.2 M. The Kb for ammonia is 1.8 x 10-5. 1.0 moles of NH4Cl is placed in the 500 mL of ammonia solution. Assuming the volume does not change by the addition of the NH4Cl, what is the pH of the buffer solution?
So, first we need the Ka of the acid. So we use the Ka/Kb relationship to determine the Ka.
Then, we determine the molarity of the NH4Cl solution:
Now, for the base we have a concentration of 1.2 M and the acid is 2.0 M. by putting these numbers and the Ka, we get:
It is also possible to engineer a buffer of a specific pH. For instance, consider this example:
b) We need a buffer that has a pH of 4. We are using HC2H3O2 (Ka = 1.8 x 10-5) and NaC2H3O2. We have a 0.2 M solution of HC2H3O2. What concentration of NaC2H3O2 would need to be added to make the right pH buffer?
In this case, we have a lot of the information we need. We have the Ka, the desired pH and the molarity of acid. If we put these things in the equation, we will have:
By doing a little algebra, we quickly get to this point:
Now, we can 10x both sides to get:
Then, we solve for the base: