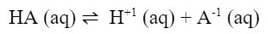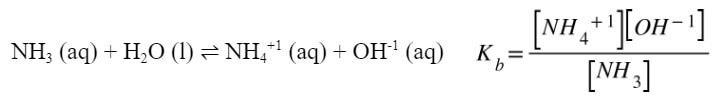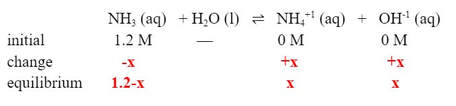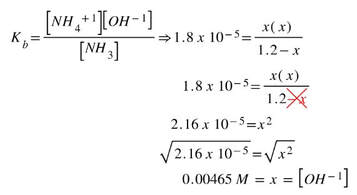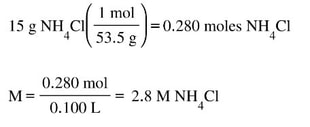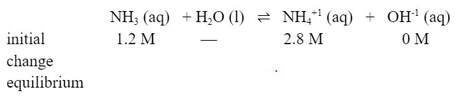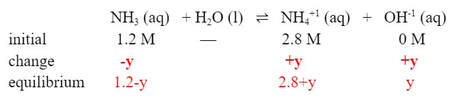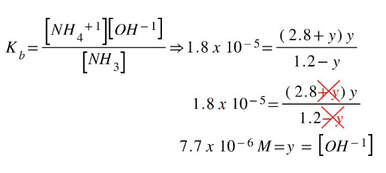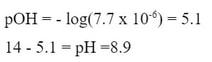Common Ion Effect
As we have already talked about, a weak acid will dissociate into a solution into its conjugate base and a hydrogen ion, as shown in the following reaction:
The Ka determines how much it will dissociate, putting some H+1 ions in the solution and some A-1 ions into the solution. Once the equilibrium is established though, what if you add more A-1 ions into the solution. According to Le Chatelier, the reaction would reverse and the products would go down to reestablish equilibrium:
Well, this would decrease the amount of H+1 ions, which would increase the pH. This is called the common ion effect. In this situation, the addition of a salt (LiA, for example), will decrease the dissociation of the acid (HA) which reduces the amount of H+1 ions in solution, changing the pH.
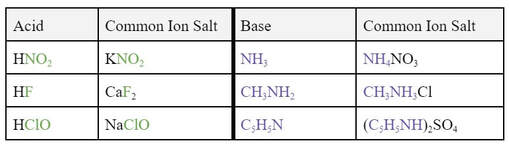
In order for the common ion effect to work, the strong electrolyte salt you add to the acid has to have the acid's conjugate base in it. So, here are a few examples:
Notice that I included bases and some of their conjugate acid salts. In the case of bases, the common ion effect would lower the pH. This is because the addition of the salt would put more of the conjugate acid in the solution, pushing the equilibrium back toward the base, reducing the amount of OH-1 in the solution. Also, notice that the conjugate acid for the base has an additional H+1 in its formula. This is because conjugate acids to bases have an addition H+1.
Calculations with Common Ion Effect
Often, when dealing with weak acids and bases, there are a mixture of acids with their common ion salts. Or bases with their common ion salts. This will change the pH of the solution. Let's do a problem to show you how it will affect the pH.
Example:
Often, when dealing with weak acids and bases, there are a mixture of acids with their common ion salts. Or bases with their common ion salts. This will change the pH of the solution. Let's do a problem to show you how it will affect the pH.
Example:
a) A solution of ammonia is made so that it has a concentration of 1.2 M. The Kb for ammonia is 1.8 x 10-5. What is the pH of the solution?
b) 15 grams of NH4Cl is placed in 100 mL of the ammonia solution. What is the new pH of the solution?
b) 15 grams of NH4Cl is placed in 100 mL of the ammonia solution. What is the new pH of the solution?
For part A, we need an equation and Kb expression:
Then, we fill in an ICE table.
Solve for x by putting it into the expression:
Since the x is the hydroxide concentration, we solve for pOH, then pH.
Now, for Part B. First, we need to calculate the concentration (Molarity) of the conjugate acid salt:
Now, this will change the ICE table. Instead of the initial [NH4+1] starting at O M, it will start at 2.8 M:
The base will still dissociate, but by a different amount. So let's call the change "y":
Solve for y by putting it into the expression: (We can take out the +y and -y, because y will be so small)
Since the y is the new hydroxide concentration because of the common ion, we solve for pOH, then pH.