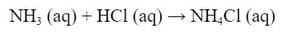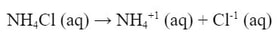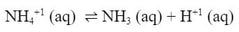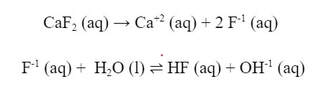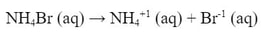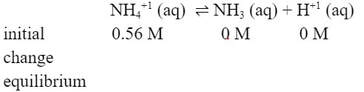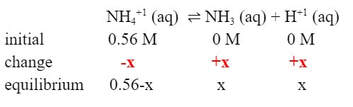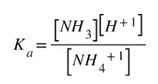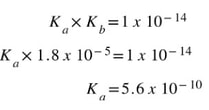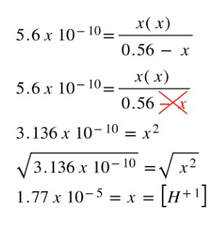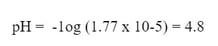pH of Salts
Why do salts have a pH other than 7?
The answer is not what you think. As we have discussed, the salt made from reacting a strong acid with a weak base will be acidic. And the salt made from mixing a weak acid with a strong base will be basic. So, you may think that the strong acid/weak base salt is acidic because the acid is stronger than the salt, which makes it acidic. Unfortunately, that's not the reasoning. It's because of the weak base.
So, why would a salt that was originally made from a weak base make the solution acidic. It's because the salt that is made from the weak base/strong acid combination contains the conjugate acid of the weak base! Here's an example of a neutralization reaction involving a weak base:
The answer is not what you think. As we have discussed, the salt made from reacting a strong acid with a weak base will be acidic. And the salt made from mixing a weak acid with a strong base will be basic. So, you may think that the strong acid/weak base salt is acidic because the acid is stronger than the salt, which makes it acidic. Unfortunately, that's not the reasoning. It's because of the weak base.
So, why would a salt that was originally made from a weak base make the solution acidic. It's because the salt that is made from the weak base/strong acid combination contains the conjugate acid of the weak base! Here's an example of a neutralization reaction involving a weak base:
The reaction produces the salt NH4Cl. This salt (like all salts) is a strong electrolyte which completely dissociates in water:
Wait! NH4+1 is the conjugate acid to NH3. Because it is a weak acid, it will partially dissociate into water:
And now we have H+1 ions in the water which will lower the pH. The Cl-1 has no acid/base properties because it came from a strong acid, so it does not influence the pH. Since the only thing in the water is a weak acid, the pH goes down.
This happens too with a strong base/weak acid salt like CaF2. The salt dissociates into Ca+2 and F-1, and the F-1 is a weak conjugate base to the weak acid HF, so it reacts with water to form OH-1 ions:
This happens too with a strong base/weak acid salt like CaF2. The salt dissociates into Ca+2 and F-1, and the F-1 is a weak conjugate base to the weak acid HF, so it reacts with water to form OH-1 ions:
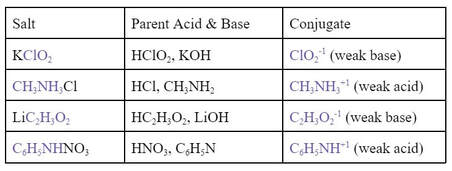
This means that salts have their own pH away from seven because of the weak acids or weak bases within them. Below are some examples of salts, their parent acids and bases, and the conjugate that causes the pH to be something other than 7.
Determining the pH of a Salt
Now that you know why, we can use this information to determine the pH of a salt. We will do one example for an acidic salt and one example for a basic salt.
Example 1:
Now that you know why, we can use this information to determine the pH of a salt. We will do one example for an acidic salt and one example for a basic salt.
Example 1:
What is the pH of a 0.56 M solution of NH4Br?
So, first, we write the dissociation of the salt, which is a strong electrolyte:
The concentration of the NH4+1 is 0.56 M. It is a weak acid (the conjugate acid of NH3). Writing the equation for the dissociation of the weak acid and the initials for the ICE table:
The [NH4+1] has to go down by some amount (-x) as it dissociates, while the [NH3] and [H+1] go up by the same amount (+x):
We can write a Ka expression for the dissociation.
Now, the Ka for NH4+1 is not listed in the back of the book. But, the Kb for NH3 is 1.8 x 10-5. So, we can figure out the Ka for the conjugate acid:
Then, we can use the Ka and solve for the x, which will equal the [H+1]:
Then, solve for the pH: