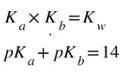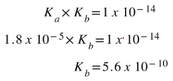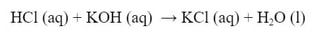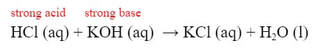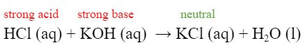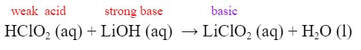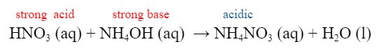Ka and Kb Relationship
Conjugate Acid & Base Relationship
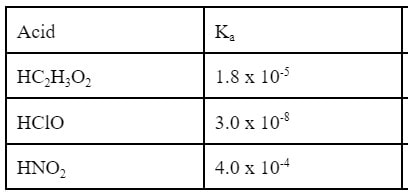
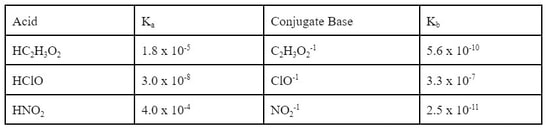
We have already learned about the conjugate relationship of acids and bases. One application of this relationship is that the Ka of an acid is related to the Kb of its conjugate base. Here's what I mean. In the back of the textbook, there is a list of common acids and their Ka values. Here is a list of some of those acids:
We have already learned about the conjugate relationship of acids and bases. One application of this relationship is that the Ka of an acid is related to the Kb of its conjugate base. Here's what I mean. In the back of the textbook, there is a list of common acids and their Ka values. Here is a list of some of those acids:
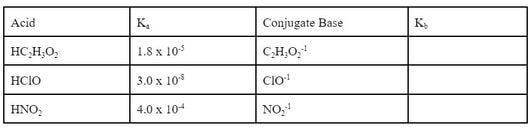
Now, each of these acids have a conjugate base. By removing an H+1 from each acid, you get the conjugate base.
Now, since each of these conjugates are weak bases, they have a Kb. However, if you go to the back of the book, you will not find any of these Kbs listed. That's because there is an easy relationship between the Ka and the Kb of the conjugates. If you know one of the two, the other can be determined by using one of the following equations:
So, for acetic acid, we can calculate the Kb for the acetate ion:
Similarly, if we do the same calculations for the other 2 acids we get:
Acid - Base Properties of Salts
Not all salts are neutral. When you think of a neutralization reaction, you think that by adding an acid to a base you would get salr and water that is neutral. However, that is not always the case. Acid-base neutralizations always make water and salt. The water is always neutral, but the salt may be neutral, or it could be acidic, or it could be basic. It all depends on the strength of the parent acid and parent base that make the salt.
For instance, KCl is a salt. It is made by combining HCl and KOH in a neutralization. The reaction would be:
Not all salts are neutral. When you think of a neutralization reaction, you think that by adding an acid to a base you would get salr and water that is neutral. However, that is not always the case. Acid-base neutralizations always make water and salt. The water is always neutral, but the salt may be neutral, or it could be acidic, or it could be basic. It all depends on the strength of the parent acid and parent base that make the salt.
For instance, KCl is a salt. It is made by combining HCl and KOH in a neutralization. The reaction would be:
Now, if we classify the parent acid and parent base for the reaction, we get:
When a strong acid and a strong base are added together, the salt they make is neutral:
When a weak acid is added to a strong base, when the solution is added in stoichiometric proportions, the salt produced is basic in nature.
When a strong acid is added to a weak base, when the solution is added in stoichiometric proportions, the salt produced is acidic in nature.
Breaking it down:
- Salts derived from a strong acid and a strong base is neutral.
- Salts derived from a strong base and a weak acid will have pH> 7. (basic)
- Salts derived from a weak base and a strong acid will have a pH < 7. (acidic)
- Salts derived from a weak acid and a weak base will have a pH dependent on which is greater between the Ka of the conjugate acid and the Kb of the conjugate base.
This video is review up until 3:15. Start there for new material: