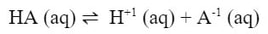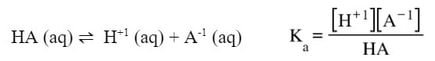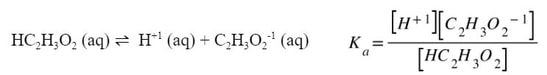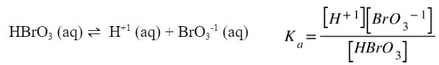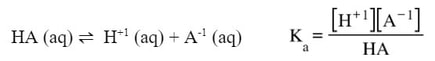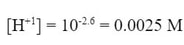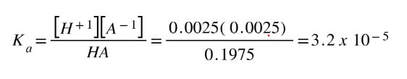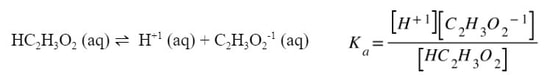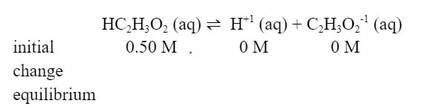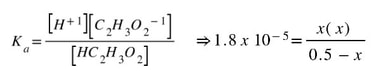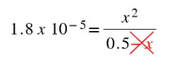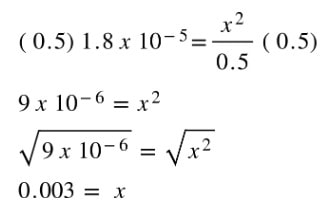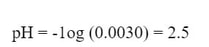Weak Acids & Ka
|
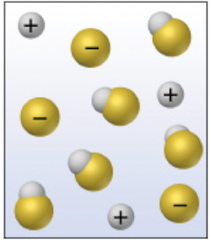
There are only 6 strong acids. There are dozens, if not hundreds, of weak acids. Weak acids are weak electrolytes that do not dissociate completely in water. Rather than completely breaking up into ions, they partially dissociate into ions and some of the acid stays together as whole molecules. From a visual standpoint, it looks a lot like the image to the right. Notice that the acid (gold-white) still has whole molecules when dissolved, but there are a few positive ions and a few negative ions.
From a chemical reaction standpoint, the main difference is that the reaction is written as an equilibrium. A generic version would look like: |
This chemical reaction is written as an equilbrium because all species exist at the same time. The forward and reverse reaction are happening at the same rate, so it indicates that all the materials are there. Since this is an equilibrium, we can write a K expression for it:
Notice in the above example that since all materials are dissolved in water, or aqueous, they all need to be included into the K expression. Since this K expression deals with an acid, instead of using Kc, we will use Ka, which stands for the acid dissociation constant. Each weak acid has a dissociation and a Ka. For example:
Now, because these are constants, that means each acid has its own value for Ka. These values can be looked up in any chemistry textbook or found online. One such list is here.
Calculations involving Ka
There are several types of calculations that can be done involving the Ka. We will show a couple of those here:
#1) Determining the Ka of an acid from its initial concentration and the pH of the solution:
Example: A 0.20 M unknown weak monoprotic acid solution has a pH of 2.6. What is its Ka?
Solution:
First we write a dissociation equation and Ka expression for this unknown acid. Since we do not know the anion of the acid, we will simply call in HA:
There are several types of calculations that can be done involving the Ka. We will show a couple of those here:
#1) Determining the Ka of an acid from its initial concentration and the pH of the solution:
Example: A 0.20 M unknown weak monoprotic acid solution has a pH of 2.6. What is its Ka?
Solution:
First we write a dissociation equation and Ka expression for this unknown acid. Since we do not know the anion of the acid, we will simply call in HA:
Since we know the pH, we can undo the pH to determine the concentration of H+1 ion at equilibrium (once the max has ionized):
This number is the equilibrium concentration of H+1. The initial amount of [H+1] was zero, and the concentration of [HA] at the beginning was 0.20 M. So, we can fill in parts of the ICE table for the reaction:
Then, we can use the differences between the initials and the finals to fill in the rest of the table:
Then, using the equilibrium values, we can calculate the Ka
#2) Determining the pH of a known concentration acid using its Ka.
What is the pH of a 0.5 M acetic acid solution?
To do this, we also need a chemical reaction for the dissociation of the weak acid and a Ka expression:
What is the pH of a 0.5 M acetic acid solution?
To do this, we also need a chemical reaction for the dissociation of the weak acid and a Ka expression:
Now, we know that the initial concentration of HC2H3O2 is 0.50 M. We also know that the initial concentration (before the acetic acid starts dissolving) of [H+1] [C2H3O2-1] are both 0 M:
However, we do not know how much the reactant dissociates, nor do we know how many ions are going to be in solution at equilibrium. We do know, though that the amount of HC2H3O2 has to go down by some unknown amount (-x) and the amount of H+1 and C2H3O2-1 both have to go up by that same unknown amount (+x). That fills in the rest of the ICE table to look like this:
We can look up the Ka for acetic acid in the back of the textbook or online to see that Ka for HC2H3O2 is 1.8 x 10-5. Using this as the Ka, and filling in the numbers at equilibrium, we get:
From here, we can solve for x. However, once we get into this we will have to solve a quadratic equation as written. But, we can make the math easier. For the 0.50 - x for the acetic acid concentration, whatever we get for x is going to be significantly smaller than 0.50. How significant? Well, as you will see in a second, the value of x we get will not change 0.50 M (to sig figs) by subtracting it. Therefore, we can omit the -x from the math:
The algebra becomes significantly easier, and we can solve for x:
x is equal to the [H+1] at equilibrium, so we can use this number and convert to pH.