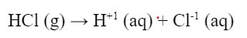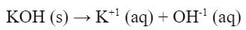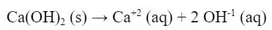Strong Acids and Bases
Acids and bases are defined by what ions they put into water. Acids dissociate to put H+1 ions into the water. Bases dissociate to put OH-1 ions into the water. When this happens, just like other substances, the acids and bases can be strong or weak electrolytes. When strong acids dissolve in water, they completely dissociate into ions, as shown in the equation:
There are two really important things to what we need to know from this.
1) The mole ratio of HCl to H+1 is 1:1. Which means the concentration (Molarity) of the acid is equal to the concentration of H+1.
2) There is no more HCl in the solution. It is only H+1 and Cl-1 ions.
All of what was said about strong acids applies to strong bases as well. The difference is that we are talking about OH-1 ions instead.
1) The mole ratio of HCl to H+1 is 1:1. Which means the concentration (Molarity) of the acid is equal to the concentration of H+1.
2) There is no more HCl in the solution. It is only H+1 and Cl-1 ions.
All of what was said about strong acids applies to strong bases as well. The difference is that we are talking about OH-1 ions instead.
Also, with bases, there are some strong bases that have 2 OH-1 ions in them, so the reaction looks like:
And in this case, the OH-1 concentration would be twice the Ca(OH)2 concentration because the mole ratio of Ca(OH)2 to OH-1 is 1:2.
As a quick reminder, here is the list of strong acids and strong bases:
As a quick reminder, here is the list of strong acids and strong bases:
|
Strong Acids: (SPN a BRIC)
sulfuric acid - H2SO4 perchloric acid - HClO4 nitric acid - HNO3 hydrobromic acid - HBr hydroiodic acid - HI hydrochloric - HCl |
Strong Bases: (hydroxide with alkali and alkali earth metals)
LiOH NaOH KOH RbOH CsOH Be(OH)2 Mg(OH)2 Ca(OH)2 Sr(OH)2 Ba(OH)2 |