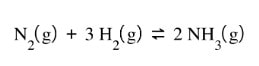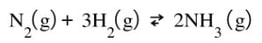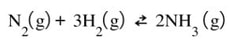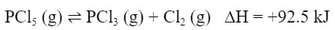Le Chatelier's Principle
We have now discussed, at length, the applications of equilibrium. But, what if you have a system that is happily at equilibrium, all is good with the world, and then some mean chemist (or chemistry student) comes in an messes up that equilibrium? Meaning, the nasty hobbit goes and adds more of the reactant, or changes the pressure of a product, or increases the temperature of the system. What does that do to the equilibrium? Well, obviously it throws it out of equilibrium and the system attempts to return itself to equilibrium. This predicted resulting change trying to get back to equilibrium is called Le Chatelier's Principle, developed by one of the greatest celebrities of the French physical chemists of the early 20th C., Henri Louis Le Chatelier.
Le Chatelier's Principle has 3 ways of looking at alterations to an equilibrium: A change of concentration one of the materials in the equilibrium, a change in pressure of the system, or a change in temperature of the system. These are the the 3 ways you can change the equilibrium and cause it to shift. Let's look at one at a time.
1) Change in concentration
Let's say you have the following reaction at equilbirum:
Le Chatelier's Principle has 3 ways of looking at alterations to an equilibrium: A change of concentration one of the materials in the equilibrium, a change in pressure of the system, or a change in temperature of the system. These are the the 3 ways you can change the equilibrium and cause it to shift. Let's look at one at a time.
1) Change in concentration
Let's say you have the following reaction at equilbirum:
If the system is at equilibrium, that means that you have the perfect amount of nitrogen, hydrogen and ammonia. The system has set itself up so that the forward and reverse reaction are happening at the same rate, keeping the amount of all components the same.
Well, what if you go and add more nitrogen to the container. That is going to disrupt the equilibrium - meaning, now you have too much nitrogen, and not enough ammonia to keep the equilibrium constant the same. The reaction is "unbalanced", or out of equilibrium. Because this reaction sets up an equilibrium, it is going to automatically do what it can to get back to equilibrium. In order to get back to equilibrium, the nitrogen will need to go down and the ammonia will need to go up. So the reaction will shift to the right and make more products.
Well, what if you go and add more nitrogen to the container. That is going to disrupt the equilibrium - meaning, now you have too much nitrogen, and not enough ammonia to keep the equilibrium constant the same. The reaction is "unbalanced", or out of equilibrium. Because this reaction sets up an equilibrium, it is going to automatically do what it can to get back to equilibrium. In order to get back to equilibrium, the nitrogen will need to go down and the ammonia will need to go up. So the reaction will shift to the right and make more products.
At some point, the ammonia will go up enough and the nitrogen will go down enough to get back to the necessary ratio and reestablish equilibrium.
You can also disrupt the equilibrium by removing something. Let's say, same equation, you remove some of the hydrogen when it is at equilibrium. That means, there is not enough hydrogen to keep the ratio the same. The ammonia will start breaking down to create more hydrogen to compensate, and the reaction will shift left:
Le Chatelier's Principle is simply using the idea that the reaction wants to get back to equilibrium when it is thrown off. The purpose of the principle is to be able to predict how it gets back to equilibrium. Bottom line:
2) Change in Pressure
Same reaction, at equilibrium.
- adding more of a material will push the equilibrium away from that substance, as it will use up the added material.
- taking out a material will push the equilibrium toward that material in order to make more of what was removed.
2) Change in Pressure
Same reaction, at equilibrium.
Now, let's say we change the pressure. The easiest way to do this is to think that increasing pressure can be done by decreasing the volume. You shrink the size of the container, the pressure is going to go up. That is going to mess up the equilibrium of gases because the pressure will not change proportionally and the Kp will be thrown off.
Bottom line:
3) Change in Temperature
This will apply to chemical reactions that are affected by heat. Meaning, if you have an exothermic or endothermic reaction. Suppose you have this reaction:
- An increase in pressure will decrease volume, which will shift the equilibrium toward the side of the reaction with the smaller number of moles.
- An decrease in pressure will increase volume, which will shift the equilibrium toward the larger number of moles.
3) Change in Temperature
This will apply to chemical reactions that are affected by heat. Meaning, if you have an exothermic or endothermic reaction. Suppose you have this reaction:
A chemical reaction with a -ΔH is an exothermic reaction, which is a reaction that releases heat. A chemical reaction with a +ΔH is an endothermic reaction, which is a reaction that absorbs heat. ΔH is always listed for the forward reaction. The above reaction is an endothermic reaction going forward, but the reverse reaction is exothermic.
By changing the temperature of the reaction, the chemical reaction is going to be forced out of equilibrium because you are changing the heat involved. In the above reaction, if we were to increase the temperature that the reaction takes place at, there will be more available heat for the reaction to absorb. The reaction will absorb some of the excess heat and move in an endothermic reaction, which means it will move forward, creating more products.
If we were to cool the reaction flask, the reaction would then start making heat to compensate. In order for this to happen, the exothermic reaction would begin to occur, which in the case above would be reverse, or making reactants.
If we were to cool the reaction flask, the reaction would then start making heat to compensate. In order for this to happen, the exothermic reaction would begin to occur, which in the case above would be reverse, or making reactants.