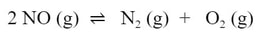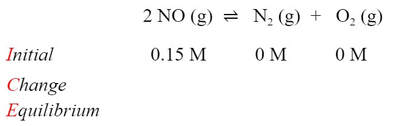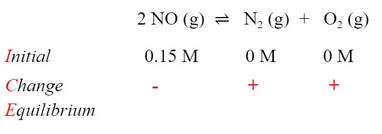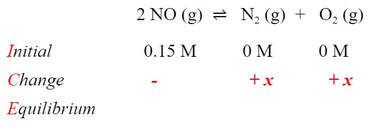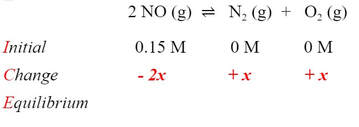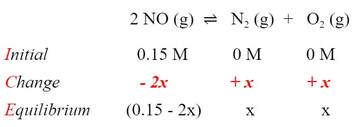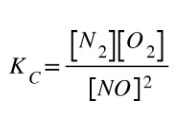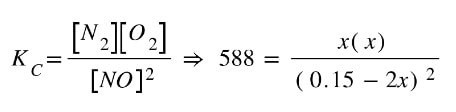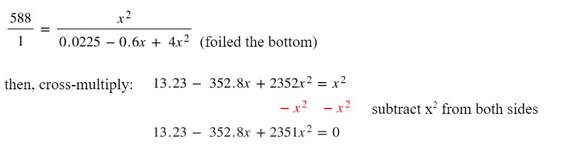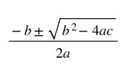Applications of Equilibrium
ICE Problem #2
The last type of equilibrium constant problem we like to give is one where you are given all the initial concentrations for the mixture and the Kc or Kp, and then ask for the equilibrium concentrations. This one is exceptionally tricky, because you have to use the skills we learned in the previous ICE problem, but with "x" instead of the changes. Again, this is easier to see as an example:
The last type of equilibrium constant problem we like to give is one where you are given all the initial concentrations for the mixture and the Kc or Kp, and then ask for the equilibrium concentrations. This one is exceptionally tricky, because you have to use the skills we learned in the previous ICE problem, but with "x" instead of the changes. Again, this is easier to see as an example:
Suppose you are dealing with this reaction:
And you were given this information:
The KC for the reaction above at 2300 K is 588. If a flask that is injected with nitrogen monoxide gas (NO) to a 0.0042 M concentration, what will be the equilibrium concentration of all the materials?
The plan for solving this problem is to do another ICE problem, putting in all the initial concentrations and finding the equilibrium concentration of all three materials. Writing the reaction and filling in the concentrations of reactant given and zero molarity for the products (we can assume this because it does not mention that this was added), we get:
Now, we know that the products have to increase, because at zero M, there needs to be some of the reactants to get to an equilibrium constant above zero. In order for the products to increase, the reactant would have to go down to form some products. So, the signs of the change have to be negative for the reactant and positive for the products:
Unfortunately, we don't know how much the N2 or O2 have to go up. They are going to go up from zero by some "unknown" amount. We can use this to our advantage, because we can say that the N2 and O2 concentration both have to go up by "x"
Which means that the NO concentration has to go down by "2x", because for every mole change of N2 and O2, there is a 2 mole change in NO:
By adding and subtracting the changes from the initials, we can fill in the equilibrium concentrations in the table:
Now, if we write the Kc expression for the reaction
We can then plug in the equilibrium concentrations that involve "x" into the equation, as well as the Kc value:
At this point, the chemistry is mostly done. We need now to do some fairly complex algebra to simplify and set one side of the equation to zero.
Now that we have the equation solved for zero, we can use the quadratic equation:
And input the a, b and c from the equation into the quadratic equation and solve:
When solving for the quadratic equation, you get 2 answers. But there cannot be two answers. That won't work. Fortunately, when you do this, one answer will not work. In this case, the value 0.0764 is too big to be subtracted from 0.15 M - it would give a negative value for the concentration of NO. Since the lowest possible value for concentration is zero, we can eliminate that value. Hence, the only possible answer for "x" is 0.0735.
Now, we haven't really answered the question yet though. The question asks for the equilibrium concentrations of all the materials. So, we need to plug our "x" into the equilibrium values from the ICE table. Thus, we get:
Now, we haven't really answered the question yet though. The question asks for the equilibrium concentrations of all the materials. So, we need to plug our "x" into the equilibrium values from the ICE table. Thus, we get: