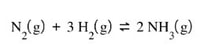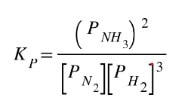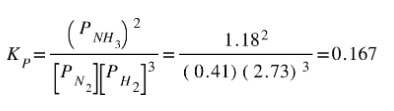Applications of Equilibrium
ICE Problem #1
Another type of equilibrium constant problem we like to give is one where you are given all the initial concentrations for the mixture, and then one of the equilibrium concentrations. Then, you'll be asked to determine the Kc or Kp. In order to do that, you have to stoichiometrically determine all the rest of the equilibrium concentrations, then calculate the K value. Again, this is easier to see as an example:
Another type of equilibrium constant problem we like to give is one where you are given all the initial concentrations for the mixture, and then one of the equilibrium concentrations. Then, you'll be asked to determine the Kc or Kp. In order to do that, you have to stoichiometrically determine all the rest of the equilibrium concentrations, then calculate the K value. Again, this is easier to see as an example:
Suppose you are dealing with this reaction:
And you were given this information:
A flask that is filled with hydrogen gas (H2) to give a pressure of 4.5 atm has enough nitrogen (N2) pumped into to bring the partial pressure of nitrogen to 1.0 atm and allowed to react to form ammonia gas (NH3). When the reaction reaches equilibrium, the partial pressure of ammonia is 1.18 atm. What is the Kp for the reaction?
In order to do this, we need to learn a new technique. It is called doing an ICE problem. ICE is an acronym for Initial, Change, Equilibrium. Essentially what we do is create a table to fill in underneath the reaction. For this problem, it would look like this:
Initial means the initial, or starting, concentrations or pressures. Next to the word initial, we will fill in the starting pressures given:
And, since there was no ammonia (NH3) put into the reaction, we can assume that the starting pressure of ammonia is 0 atm:
Now, the one other piece of information we know is that the equilibrium pressure of ammonia is 1.18 atm. So, we can put that in as the equilibrium pressure of ammonia:
In terms of change, the ammonia went from 0 to 1.18 atm, so it increased by +1.18 atm. We can put that in for the change in ammonia:
Stoichiometry can allow us to figure out the rest of the changes. Since ammonia went up by 1.18 atm, the nitrogen would have had to have gone down. Since the ratio is 1 nitrogen for every 2 ammonia, then there would be -0.59 atm of nitrogen needed to make 1.18 atm of ammonia, or half as much:
Likewise, if nitrogen went down, so would the hydrogen. The ratio of nitrogen to hydrogen is 1:3, so a -0.59 atm of nitrogen would mean a (3 x -0.59 atm) or -1.77 atm change for hydrogen:
From here, we can subtract the initial minus the change, and get the equilibrium pressures:
If we take the original equation, we can determine the Kp expression:
And then plug in the equilibrium pressures: