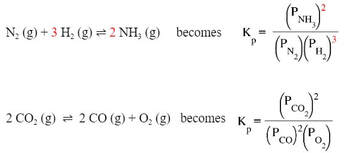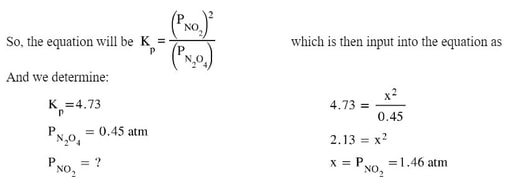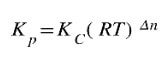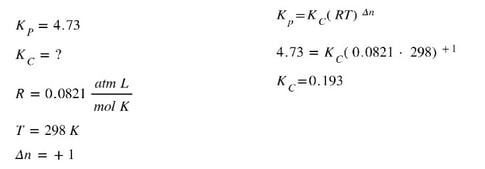Equilibrium Constant
Equilibrium Constant for Pressure (Kp)
Sometimes, when measuring equilibrium, it is easier to measure the partial pressures of the gases involved as opposed to their molarities. The nice part is that equilibrium pressures will be proportional to the molarities, so the equilibrium constant will be calculated in the same way. So, if you have a generic equation like this:
Sometimes, when measuring equilibrium, it is easier to measure the partial pressures of the gases involved as opposed to their molarities. The nice part is that equilibrium pressures will be proportional to the molarities, so the equilibrium constant will be calculated in the same way. So, if you have a generic equation like this:
Just like before, the products are on top, the reactants on the bottom. The coefficients (indicated in red) become exponents for the substances in the equilibrium expression. The main difference is that you use the partial pressures of the gases in the mixture. Atmospheres instead of molarity is used as the measurement. A couple more examples:
Calculations using Kp
As with Kc, the Kp can be calculated in the same way. The only difference is the numbers will be in atmospheres.
Example 1:
As with Kc, the Kp can be calculated in the same way. The only difference is the numbers will be in atmospheres.
Example 1:
If the Kp for the reaction N2O4 (g) <==> 2 NO2 (g) is 4.73 at 298 K, and the equilibrium pressure of N2O4 is 0.45 atm, what is the equilibrium pressure of NO2?
Kp and Kc
There is a relationship between Kp and Kc. They can be converted from one to another. A manipulation of the ideal gas law solves for the following relationship:
There is a relationship between Kp and Kc. They can be converted from one to another. A manipulation of the ideal gas law solves for the following relationship:
where R is the universal gas constant (0.0821 atm L/mol K), T is the Kelvin temperature and Δn is the change in moles. To determine the change in moles, add the total moles of products minus the total moles of the reactants. In the equation,
N2 (g) + 3 H2 (g) <==> 2 NH3 (g)
there are a total of 4 moles on the reactants side, and two moles on the products side, so 2-4 = -2. So, Δn = -2.
Example 2:
If the Kp for the reaction N2O4 (g) <==> 2 NO2 (g) is 4.73 at 298 K, what is the Kc for this reaction at 298 K?
N2 (g) + 3 H2 (g) <==> 2 NH3 (g)
there are a total of 4 moles on the reactants side, and two moles on the products side, so 2-4 = -2. So, Δn = -2.
Example 2:
If the Kp for the reaction N2O4 (g) <==> 2 NO2 (g) is 4.73 at 298 K, what is the Kc for this reaction at 298 K?