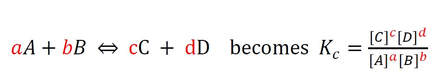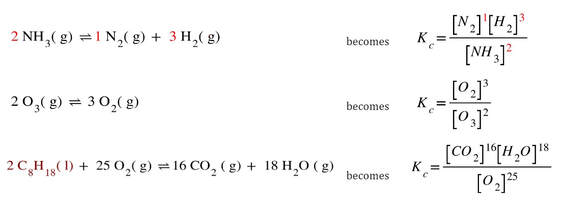Equilibrium Constant
Graphing Equilibrium
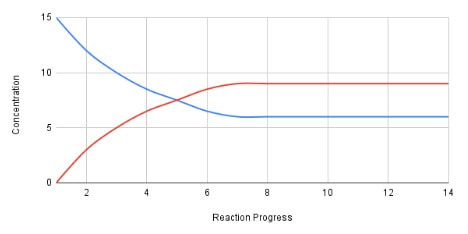
For a reversible reaction, you can measure, over time, the amount of reactant and the amount of product within the reaction flask. As you do this, if you graph the amounts versus time, an interesting phenomenon occurs. Let's take a hypothetical example of a reaction X(g) <==> Z(g). If you start with just X(g) (at a concentration of 15) in the reaction flask, and measure the amounts of X and Z every minute, you may get a graph that looks like this:
For a reversible reaction, you can measure, over time, the amount of reactant and the amount of product within the reaction flask. As you do this, if you graph the amounts versus time, an interesting phenomenon occurs. Let's take a hypothetical example of a reaction X(g) <==> Z(g). If you start with just X(g) (at a concentration of 15) in the reaction flask, and measure the amounts of X and Z every minute, you may get a graph that looks like this:
In the above example, the blue line is the amount of X and the red line is the amount of Z. As time progresses, and X turns into Z, the amount of X goes down and the amount of Z goes up. But, notice that the amount X does not drop to zero. It levels off at about 6, while the amount of Z levels off at about 9. This is because as more Z is made, some of that Z turns back into X. At some point the amount of Z and X do not change, because (and this is important), the amount of X turning into Z at any one moment is equal to the amount of Z turning into X! This is called equilibrium.
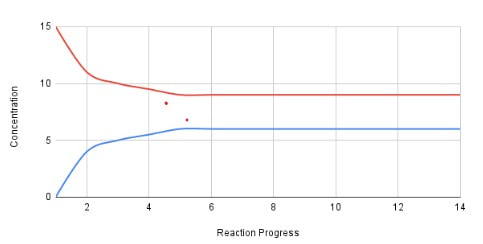
Now, look what happens in the same reaction if we start with a concentration of 15 in Z, with no X in the reaction flask:
Now, look what happens in the same reaction if we start with a concentration of 15 in Z, with no X in the reaction flask:
Again, the red line is Z and the blue line is X. The amount of Z goes down, because the reaction is reversible and Z can make X (X is going up). However, we reach a point where the two lines level out, and they level out at the same place, with Z at a concentration of 9 and X at a concentration of 6. This means, that no matter where you start, the proportion of Z:X will be the same. In this case, the proportion always ends up 9:6, or 3:2.
Chemists find that the proportionality between the reactants and products of any reversible reaction always ends up at the same place. You can start with any amount of any reactant, and the ratio of products to reactants for any reaction is always the same. It is not always 3:2, but there will be some ratio that it always ends up at. This is called an equilibrium constant. An equilibrium constant is a number that describes the ratio of the product to the reactant in any chemical reaction. Each reversible reaction has its own value for its equilibrium constant, but it is always calculated in the same way.
Equilibrium Constant
Mathematically and experimentally, it was determined that the ratio of products to reactants are dependent not only on the concentrations of the materials, but also on the balancing. Danish scientists Cato Guldberg and Peter Waage mathematically determined the relationship between the products and reactants in equilibrium. They proposed the equilibrium constant. The equilibrium constant is based on the balanced chemical reaction.
Generically, converting a balanced equation to a equilibrium constant, the process is:
Chemists find that the proportionality between the reactants and products of any reversible reaction always ends up at the same place. You can start with any amount of any reactant, and the ratio of products to reactants for any reaction is always the same. It is not always 3:2, but there will be some ratio that it always ends up at. This is called an equilibrium constant. An equilibrium constant is a number that describes the ratio of the product to the reactant in any chemical reaction. Each reversible reaction has its own value for its equilibrium constant, but it is always calculated in the same way.
Equilibrium Constant
Mathematically and experimentally, it was determined that the ratio of products to reactants are dependent not only on the concentrations of the materials, but also on the balancing. Danish scientists Cato Guldberg and Peter Waage mathematically determined the relationship between the products and reactants in equilibrium. They proposed the equilibrium constant. The equilibrium constant is based on the balanced chemical reaction.
Generically, converting a balanced equation to a equilibrium constant, the process is:
So, this process can be a little confusing. Essentially, the products are on top, the reactants on the bottom. The coefficients (indicated in red) become exponents for the substances in the equilibrium expression. So, here are a few more examples:
In the last one, you will notice that C8H18 is omitted. That is because solids and liquids do not have molarities. Only gases and aqueous can have molarities. So, we leave out any solids or liquids from the equilibrium expression. To reiterate, the equilibrium expression, K, is a different equation, based on the chemical reaction. Every reaction has its own specific K expression (the way it is calculated), and its own specific numerical value for the expression.
Couple more definitions: Homogeneous equilibria are equilibrium reactions that have all the substances within the reaction in the same state. Like #1 and #2 above. Heterogeneous equilibria have multiple states within the same reaction.