INtroduction to Equilibrium
Reversible Reactions
In chemistry, there are some reactions that only go one way. Neutralizations are an example of these. When you add acid to base, they react and form the products. However, there are some chemical reactions that can be easily reversed. In some cases, there just needs to be a simple change to make it reverse, and in some cases it is spontaneous and happens on its own.
For example, take this reaction:
In chemistry, there are some reactions that only go one way. Neutralizations are an example of these. When you add acid to base, they react and form the products. However, there are some chemical reactions that can be easily reversed. In some cases, there just needs to be a simple change to make it reverse, and in some cases it is spontaneous and happens on its own.
For example, take this reaction:
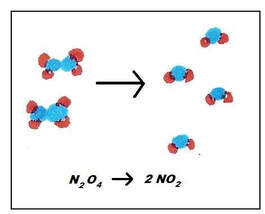
Dinitrogen tetraoxide (N2O4), a pollutant, will decompose into nitrogen dioxide (NO2) within the atmosphere. However, in the atmosphere, the reverse reaction can happen:
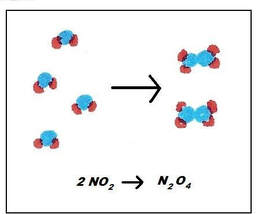
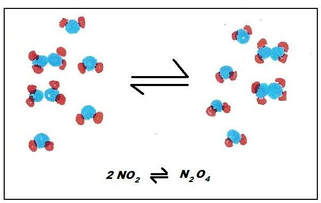
In this case, the nitrogen dioxide will combine to form dinitrogen tetraoxide. Both of these reactions are spontaneous, so these reactions can actually happen at the same time. Since the reversible reactions happen at the same time, the reaction is often written like this:
Because these two reactions happen at the same time, you never get to a point where there is only product. The reaction is always reversing, so there is always some reactant being made. But as more reactant is made, the reaction moves forward to make more product. You never end up with just reactant or just product. There is always some of both reactant and product within the reaction flask. Since the reaction perpetually goes forward and reverse, and there is always some reactant and some product, we eventually will reach an equilibrium