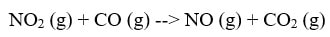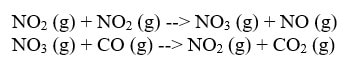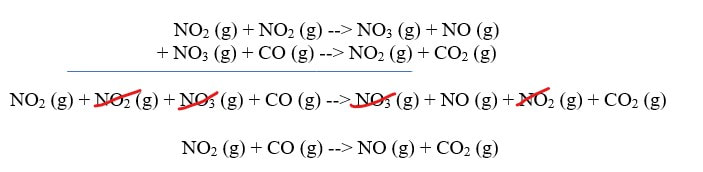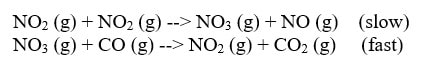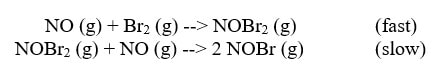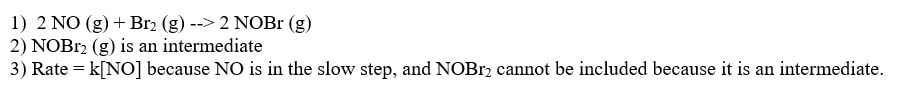Multi-Step Mechanisms
In this reaction, in order for the products to be made, one of the NO2 has to transfer an O over to the other NO2 to form NO3. Then, that NO3 can give up the O to CO to make CO2. The reaction cannot happen by simply moving an O from NO2 to CO. Now, if you use Hess's Law, you will discover that the multi-step mechanism does add up to the original reaction:
What does this have to do with rate?
Often, one of these chemical reactions within the mechanism is slower than the other. This will slow the production of product. We call the slower reaction the rate determining step. It is often shown within the mechanism something like this:
Notice that the rate law does not contain CO. Because that reaction goes fast, it doesn't affect the rate. Occasionally, the materials in the fast step will appear in the rate law, but not often.
Also, notice that NO3 is also not in the rate law. NO3 is not in the rate for two reasons:
1) NO3 is in the fast step. Fast steps do not affect the rate.
2) NO3 is also an intermediate. Intermediates are materials that are made early in a mechanism and used up by the end of the mechanism. We can never include intermediates in a rate law because we do not know the concentration of them. We did not put any NO3 in the reaction, so we do not know its concentration, so we cannot know (at any point) how much NO3 in the reaction.
1) Use Hess's Law to show the overall reaction.
2) Identify any intermediates or catalysts
3) Predict a rate law. To do this last part, you need to guess what the rate law is based on the slow step. You do not necessarily have to be correct, but it must make sense. The rate law should have at least a reactant from the slow step, but that reactant cannot be an intermediate. To do these three things for the above mechanism, you may come up with the following answer:
The real rate law based on experimentation is Rate = k[NO]2[Br2]. Although our prediction is acceptable, there must be some effect of the first reaction on the rate. In other words, the fact that NO and Br2 are needed to make the intermediate, that must slow the reaction down some, just not as much as the reaction of NOBr2 with NO.