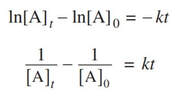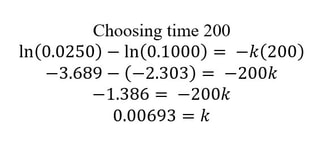An Example Using The INtegrated Rate Law
|
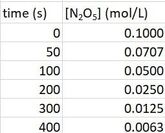
Determine the rate law for the reaction
2 N2O5 (g) --> 4 NO2 (g) + O2 (g) given the following data
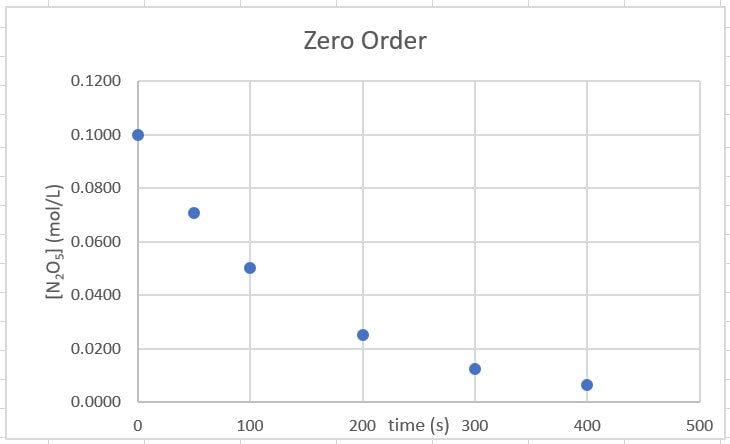
which, when plotted on a graph, will look like what is to the right:
|
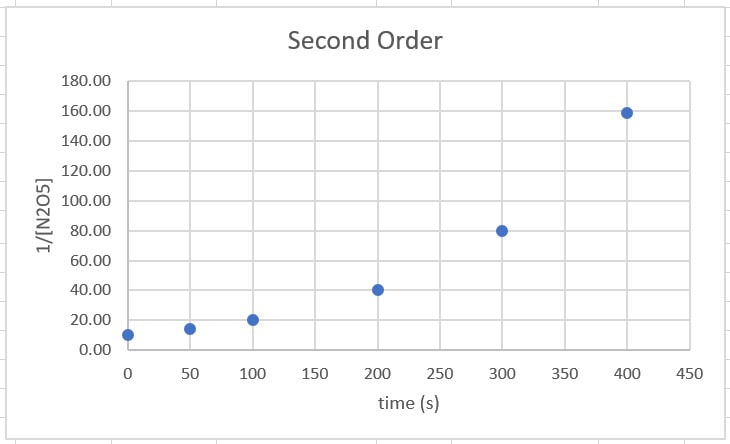
Right away, we can conclude the reaction is not zero order. When the data is plotted, it does not form a straight line, so it cannot be zero order. We need a straight line.
|
Determining Rate Order Using Graphs
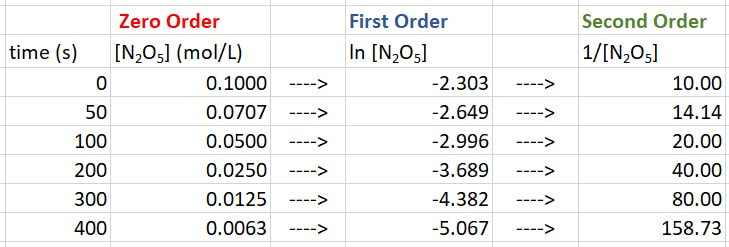
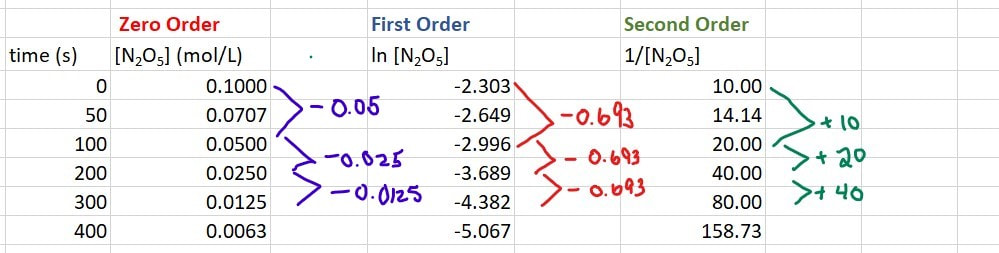
So now we need to manipulate the data. Since we do not know if this is first or second order, we will calculate both:
So now we need to manipulate the data. Since we do not know if this is first or second order, we will calculate both:
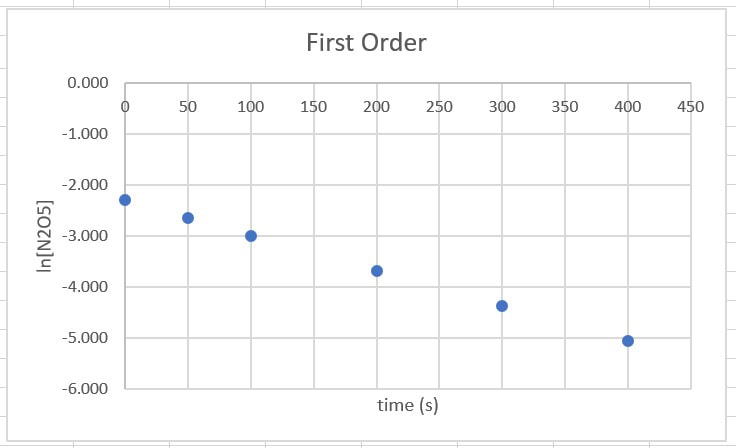
Then, we can graph both, to see which gives us a straighter line.
Now that we have both sets of data, it appears the straighter of the two lines is the first order reaction, which gives us a rate law of
Rate = k[N2O5]
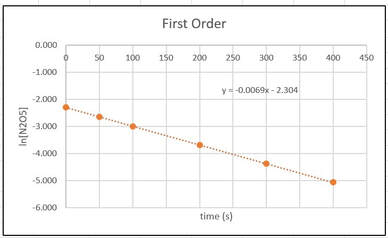
Then, if we add a trendline and trendline equation to the first order graph, we get:
From the trendline equation in the graph, we can then determine that k = 0.0069 1/s , because that is the negative slope of the reaction.
But, what if you don't have a graphing program???
This can still be figured out. You just have to look for a consistent change in number between similar data points. So, you still do the calculations for ln[conc] and 1/[conc]. We have already done that, so let's bring that down here:
But, what if you don't have a graphing program???
This can still be figured out. You just have to look for a consistent change in number between similar data points. So, you still do the calculations for ln[conc] and 1/[conc]. We have already done that, so let's bring that down here:
Now we just look at the changes in numbers for [A] (in this case N2O5), for ln[A] and for 1/[A].
For [A], from 0 - 100 s, there is a drop of 0.050, from 100 to 200 s there is a drop of 0.025, and for 200 - 300 s there is a drop of 0.0125. In each of these cases the drop is very different, so it is not zero order.
For ln[A], from 0 - 100 s, there is a drop of 0.693, from 100 to 200 s there is a drop of 0.693, and for 200 - 300 s there is a drop of 0.693. In each of these cases the drop is the same, so it is first order.
For 1/[A], from 0 - 100 s, there is a rise of 10, from 100 to 200 s there is a rise of 20, and for 200 - 300 s there is a rise of 40. In each of these cases the rise is very different, so it is not second order.
What else might be asked?
1) At this point, you may be asked to determine the rate constant. The easiest way to do this is to use one of these two equations:
For ln[A], from 0 - 100 s, there is a drop of 0.693, from 100 to 200 s there is a drop of 0.693, and for 200 - 300 s there is a drop of 0.693. In each of these cases the drop is the same, so it is first order.
For 1/[A], from 0 - 100 s, there is a rise of 10, from 100 to 200 s there is a rise of 20, and for 200 - 300 s there is a rise of 40. In each of these cases the rise is very different, so it is not second order.
What else might be asked?
1) At this point, you may be asked to determine the rate constant. The easiest way to do this is to use one of these two equations:
Click to set custom HTML
Since you know the reaction is first order, you would use the first. Now, simply pick the initial concentration, and another concentration and the time to determine k. Something like this:
2) You could be asked to determine the molarity at another unknown time, say at 150 s. Again, using the appropriate equation (the one for first order, in this case), the starting concentration and the rate constant, determine the concentration at that time:
Here are a couple more videos examples:
|
|
|