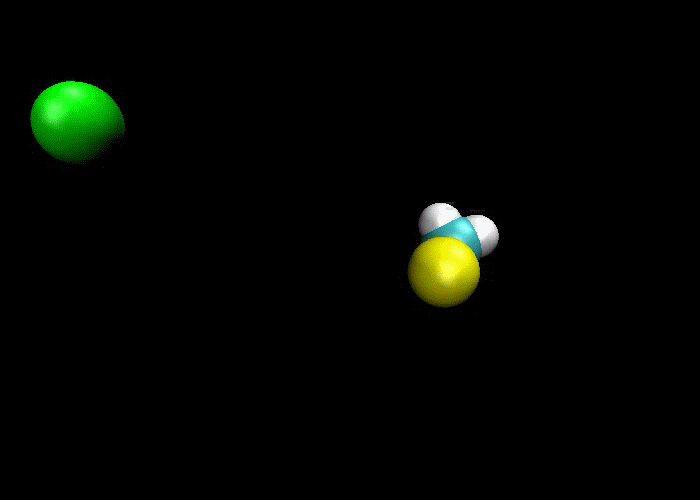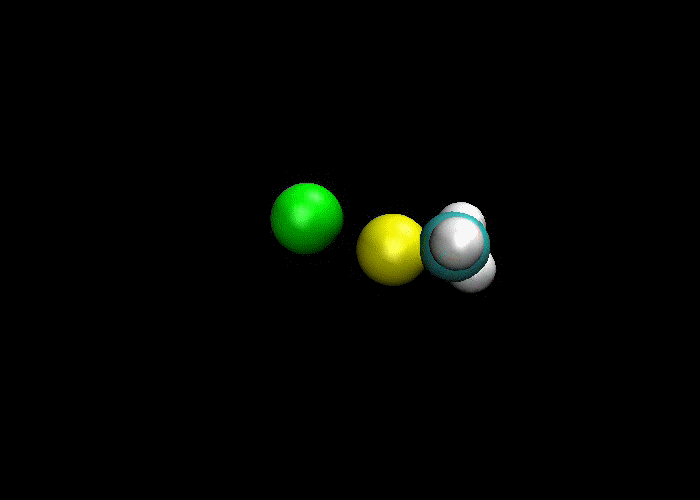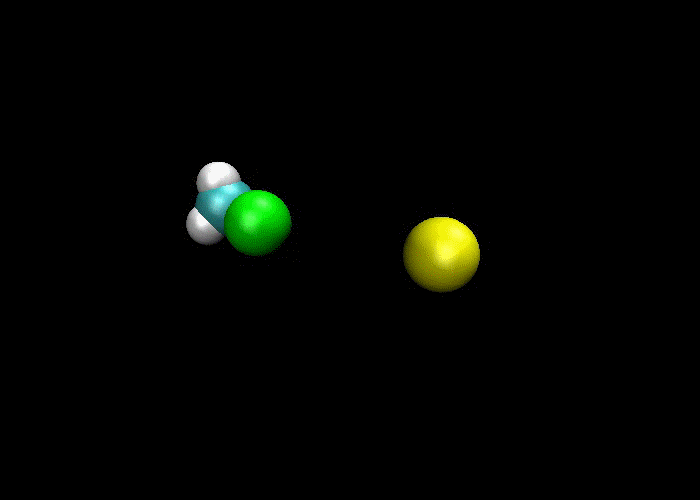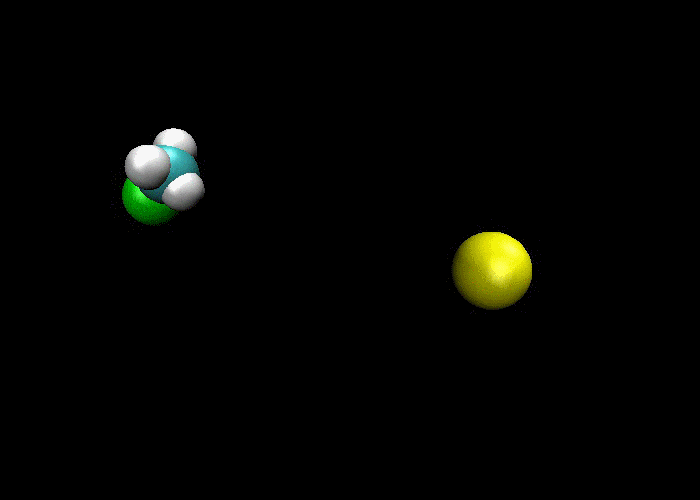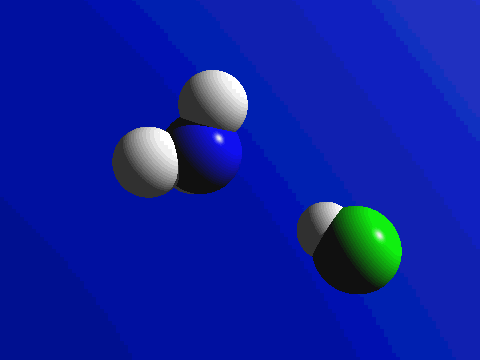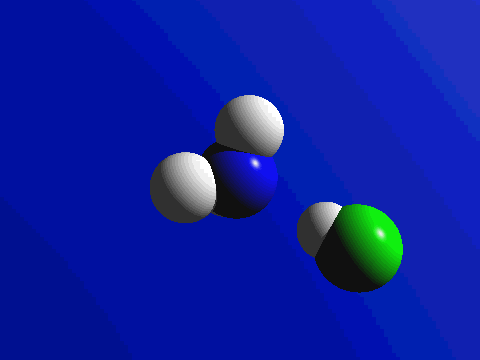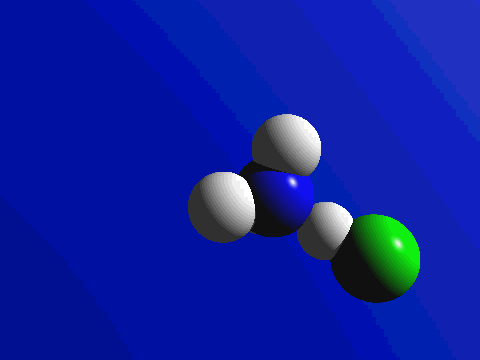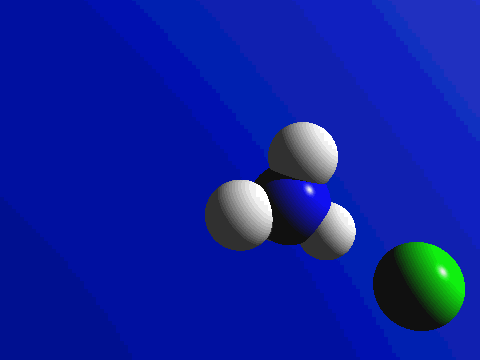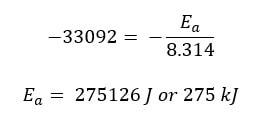Affecting the Rate of Reaction
In most cases of chemical reactions that we can measure the rates, it is experimentally found that the higher the temperature, the faster the reaction proceeds. This is not 100% true, but it is more likely than not. That being said, it must mean that the rate constant is not really constant. Because, if a chemical reaction has a certain rate law, like Rate = k[A], and you don't change the amount of A, but the rate still increases, then the k must change. Now, determining the change in k due to temperature change is WAY beyond the scope of this class. However, the reasons are not. So, let's discuss why changes in temperature would affect the k and ultimately the rate.
The Collision Model
First and foremost, we must quickly discuss why reactions happen. Knowing what we know about the kinetic molecular theory, scientists figure that in order for a reaction to occur, a collision between molecules must happen in order to transfer atoms between molecules. Below are a couple of examples of reactions where a collision transfers materials:
The Collision Model
First and foremost, we must quickly discuss why reactions happen. Knowing what we know about the kinetic molecular theory, scientists figure that in order for a reaction to occur, a collision between molecules must happen in order to transfer atoms between molecules. Below are a couple of examples of reactions where a collision transfers materials:
So, if a collision is needed in order for a reaction occurs, then if more collisions happen then more reactions will occur. This means there are a couple of ways to increase the number of collisions:
1) increase the concentration of reactants, which means more molecules present mean more collisions and more reactions.
2) increase the temperature, which makes the molecules go faster, which means more collisions would happen and more reactions.
Orientation
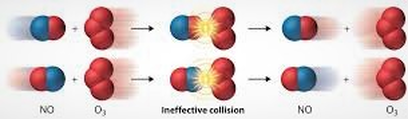
A collision may occur, but it may not be effective. Meaning, two molecules may collide, but the right part of the molecules don't hit and the reaction doesn't occur. For instance, lets take this reaction:
1) increase the concentration of reactants, which means more molecules present mean more collisions and more reactions.
2) increase the temperature, which makes the molecules go faster, which means more collisions would happen and more reactions.
Orientation
A collision may occur, but it may not be effective. Meaning, two molecules may collide, but the right part of the molecules don't hit and the reaction doesn't occur. For instance, lets take this reaction:
NO (g) + O3 (g) --> NO2 (g) + O2 (g)
Sometimes, even though the reactants collide with one another, the right atoms might not collide. In these two cases, the
In each of the cases above, they are ineffective collisions because the appropriate atoms did not touch. In the first one, the two oxygens came in contact, which doesn't transfer an oxygen. In the second, the nitrogen hits an oxygen it cannot remove because it is in the center of the molecule. In both cases, the oxygen cannot transfer. The reaction instead has to happen like below:
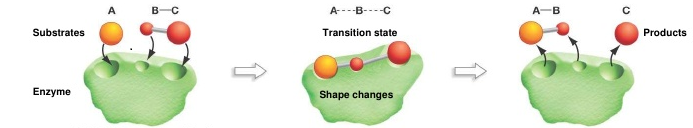
In this case, the molecules are oriented in the right way so that the nitrogen hits one of the outside oxygens of ozone to remove that oxygen. Orientation issues are often fixed by enzyme catalysts. Sometimes, catalysts can exist so that they grab onto the reactants and position them in such a way to cause the reactants to react. An example can be seen below:
Energy Profiles and Activation Energy
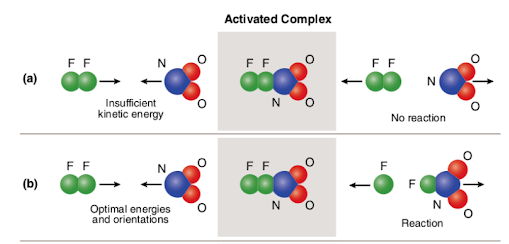
In some cases, a reaction may or may not occur because there is not enough energy in the collision to make it happen. For instance, two molecules may hit one another, and hit in the right way, but with not enough force to break the existing bonds. As you can see, in (a), the molecules collide, but there is not enough kinetic energy to allow the N to pull off one of the F.
In some cases, a reaction may or may not occur because there is not enough energy in the collision to make it happen. For instance, two molecules may hit one another, and hit in the right way, but with not enough force to break the existing bonds. As you can see, in (a), the molecules collide, but there is not enough kinetic energy to allow the N to pull off one of the F.
But, in (b), the molecules are warmer, giving the more speed and KE so that when they do collide, the collision is hard enough to knock the F loose to attach to the N. This extra energy that is needed to get the molecules is termed the activation energy, symbol Ea.
|
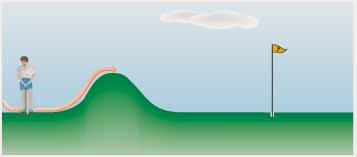
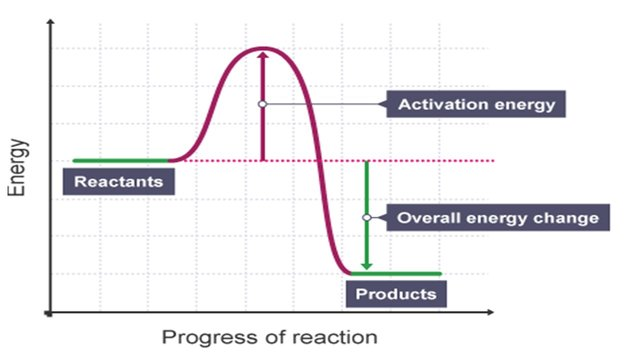
Graphically, we show the activation energy in an energy profile. The energy profile is like a miniature golf hole. If there is a hill in the way, the only way you have even a chance to get the golf ball in the hole is to hit it hard enough to get over the hill. The reactants are before the hill, and the products are after the hill. If you do not hit the ball hard enough, it does not have enough KE to get over the hill and come back to you. In this analogy, the golf ball will stay a reactant (left side of hill). But if you hit it hard enough and give it enough KE, the ball will get over the hill and become a product.
|
|
As you can see, an energy profile is very similar. The reactants are to the left, the products to the right. The bump in the middle shows the amount of energy needed in order to convert from reactants to products. The activation energy is a measure of the difference between the potential energy stored in the reactants to the top of the curve. In this case, though, once you get over the hill, the products are way below in energy compared to the reactants. That extra energy will be given off, usually as heat. In this case, you get back all the activation energy and get extra energy in the form of heat. Thus, this is going to be an exothermic reaction.
|
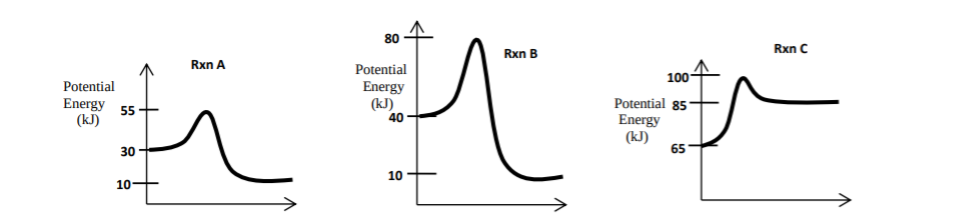
Here are three more energy profiles, this time with numbers:
In reaction A, the Ea = + 25 kJ (55 - 30 = 25), and the ΔE = - 20 kJ (10 - 30 = -20). The reaction is exothermic overall because there is a loss of energy from beginning to the end. In reaction C, the Ea = +35 kJ (100 - 65 = 35), and the ΔE = +20 kJ (85 - 65 = 20). The reaction is endothermic because there is a gain of energy from reactants to products, and you only get some of the Ea back.
|
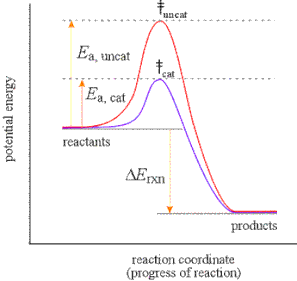
Sometimes a catalyst is used to make a reaction go faster. Sometimes the catalysts orient the reactants in such a way, that the activation energy for the collision is not as high. In these cases, there will be a change to the energy profile in which the activation is lower, or a lower bump. This can be seen in the diagram to the right.
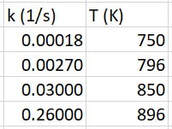
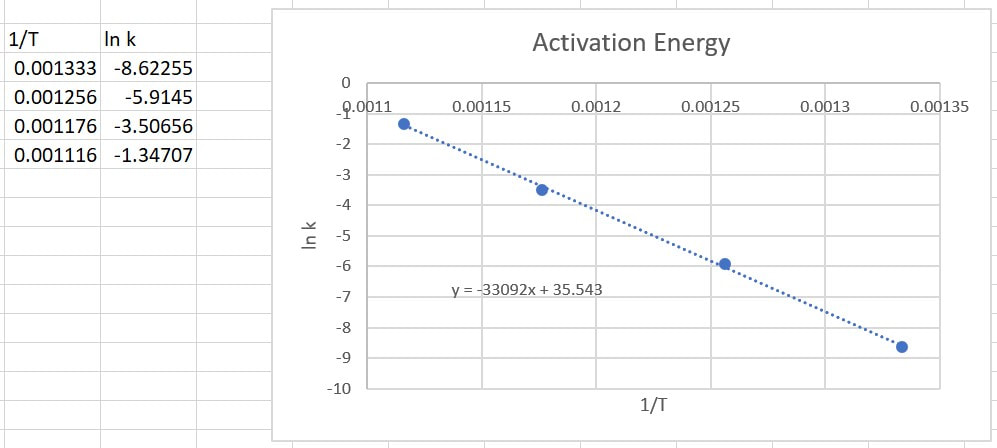
The activation energy is determined experimentally by repeating an experiment several times at different temperatures. When you change the temperature at which a reaction occurs, but nothing else, sometimes the reactions changes speed. When this happens, the rate constant must be changing. Through measurements we can get the rate constant at different temperatures. This allows us to get data that looks like this:
|
Through mathematics and relationships way beyond the scope of this course, Svante Arrhenius found a nonlinear relationship between the temperature and the rate constant. He found that if you plotted ln k versus 1/T (in Kelvin), a straight line was achieved: