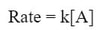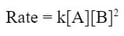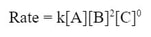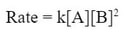Rate Law
Determination of the Rate Law
As we have seen, the speed of a chemical reaction depends on the amount of reactants you use. A reaction happens faster when there are more reactants to react. It slows down as the reactants are used up. Chemists have developed an idea of a rate law that is often developed and used to determine the speed of the reaction. Unfortunately, the rate law is not based on the coefficients from the balanced chemical reactions like equilibrium. The rate law is determined by using experimental data to figure out the law. Essentially, what you have to do is measure the rates of a reaction by performing the reaction several different times, changing the concentration of the reactants and seeing how the speed is affected. Let's do an example to show how this works.
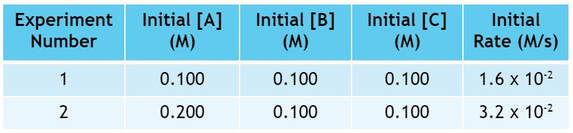
First, let's assume a generic reaction like A + B + C --> products. We perform the experiment 5 times, changing the concentration of reactants each time. Then, we measure the speed of the reaction, and get data like what you see below:
As we have seen, the speed of a chemical reaction depends on the amount of reactants you use. A reaction happens faster when there are more reactants to react. It slows down as the reactants are used up. Chemists have developed an idea of a rate law that is often developed and used to determine the speed of the reaction. Unfortunately, the rate law is not based on the coefficients from the balanced chemical reactions like equilibrium. The rate law is determined by using experimental data to figure out the law. Essentially, what you have to do is measure the rates of a reaction by performing the reaction several different times, changing the concentration of the reactants and seeing how the speed is affected. Let's do an example to show how this works.
First, let's assume a generic reaction like A + B + C --> products. We perform the experiment 5 times, changing the concentration of reactants each time. Then, we measure the speed of the reaction, and get data like what you see below:
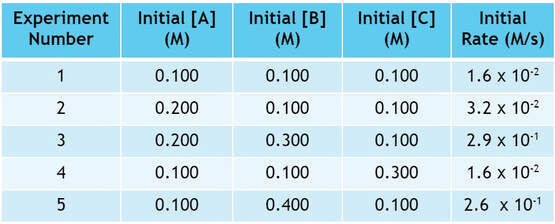
By doing these 5 experiments, we have changed the concentrations of A, B and C. As a result, the initial rates (rates at the very beginning of the reaction) have changed corresponding to the changes made in concentration. If we dissect the data, we can determine the rate law.
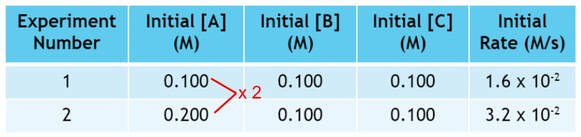
To dissect the data, you have to look at each reactant one at a time and see how it affects the rate. When we compare experiments, we must compare 2 experiments where only one factor has changed. So, we can determine how A affects the rate by looking at experiments 1 and 2:
To dissect the data, you have to look at each reactant one at a time and see how it affects the rate. When we compare experiments, we must compare 2 experiments where only one factor has changed. So, we can determine how A affects the rate by looking at experiments 1 and 2:
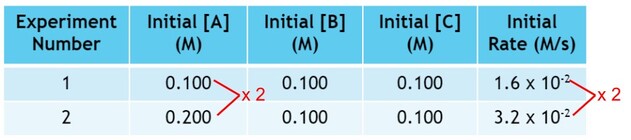
In experiment 2, we double the amount of A used from experiment 1, while keeping B and C the same:
If we look at the rate, the rate has doubled as well:
As we can see, doubling the amount of A doubles the rate. This is a one to one relationship. We can begin writing the rate law, because the rate is directly affected by the amount of A. The rate law for this reaction becomes:
[A] is the concentration of A, and k is called the rate constant. The rate constant is different for every experiment and represented by the letter k. The rate constant is the conversion factor for the experiment from a concentration to a rate.
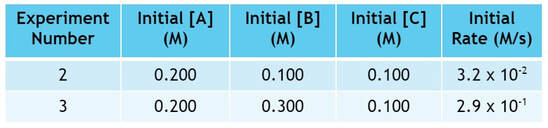
Now let's look at B. Comparing experiments 2 and 3, the amount of B changes while the amount of A and C remain constant:
Now let's look at B. Comparing experiments 2 and 3, the amount of B changes while the amount of A and C remain constant:
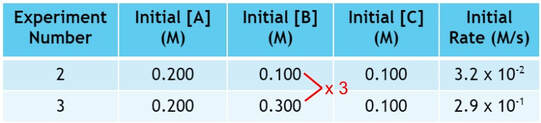
In experiment 3, we triple the amount of B used from experiment 2:
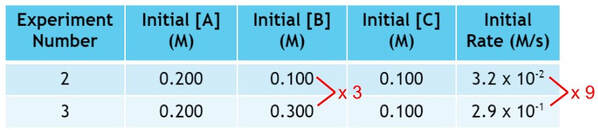
As we triple the amount of B, the rate changes by a factor of 9:
In this case, the change of B is not a 3 to 3 change. In fact tripling the amount B actually changes the rate by 9, which is 3 squared (32). Since changing B has a squaring factor on the rate, we must square B in the rate law, or:
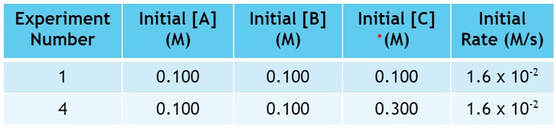
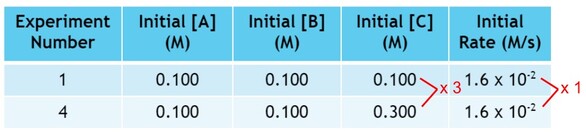
Finally, we must look at C. In looking for 2 experiments where C changes, but A & B don't. So we will compare experiments 1 and 4:
Now, in these experiments, we change C by a factor of 3, but the rate does not change:
Since, the rate is unaffected by the change in C, we can conclude that there is zero effect on the rate by the increase of C, and we can include C in the rate law to the zero power.
Or we can simply leave C out altogether: