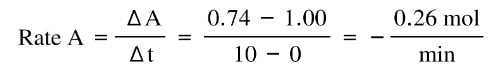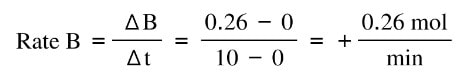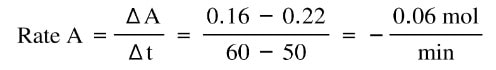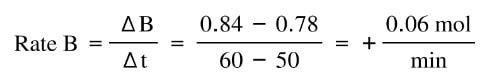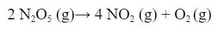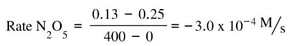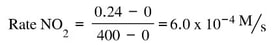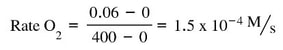Reaction Rates
Kinetics is the branch of chemistry that measures how fast a chemical reaction happens. Some chemical reactions happen really fast like the explosion of TNT, while others move really slowly like the rusting of iron. In both cases, the speed of the reaction can be measured. We call these rates of reactions, which can be affected by many things, including temperature, concentrations, surface area and catalysts. In this next unit, we will be measuring reaction rates and the factors that affect them.
|
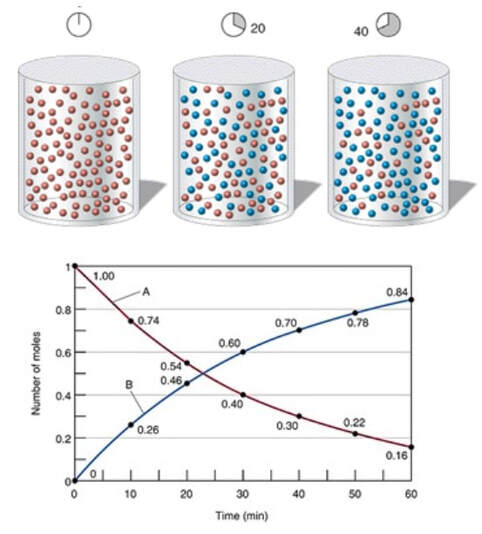
Sometimes, reactions go slow enough that you can measure the amounts of the reactants and products at different times. When you plot a hypothetical reaction like:
A --> B
At time 0, the amount of A starts going down as it is converted into B. At the same time, the amount of B increases. In the first 10 min, the rate of change of A:
And the rate of change of B is:
Notice that the change is exactly the same value, except A is negative and B is positive. That's because this reaction is one to one in terms of moles, and whatever decrease in A has to be the same as the increase of B.
|
Now, if you look at the last 10 minutes, the rates are different:
A is no longer decreasing at the same speed. Initially, the reaction was decreasing at a fast rate of -0.26 mol/min in the first 10 minutes. But as the reaction continues on longer, there is less A to react, so the reaction slows down to -0.06 mol/min in the last 10 minutes. So, the reaction does slow down over time. There are different rates depending on the amount of material present.
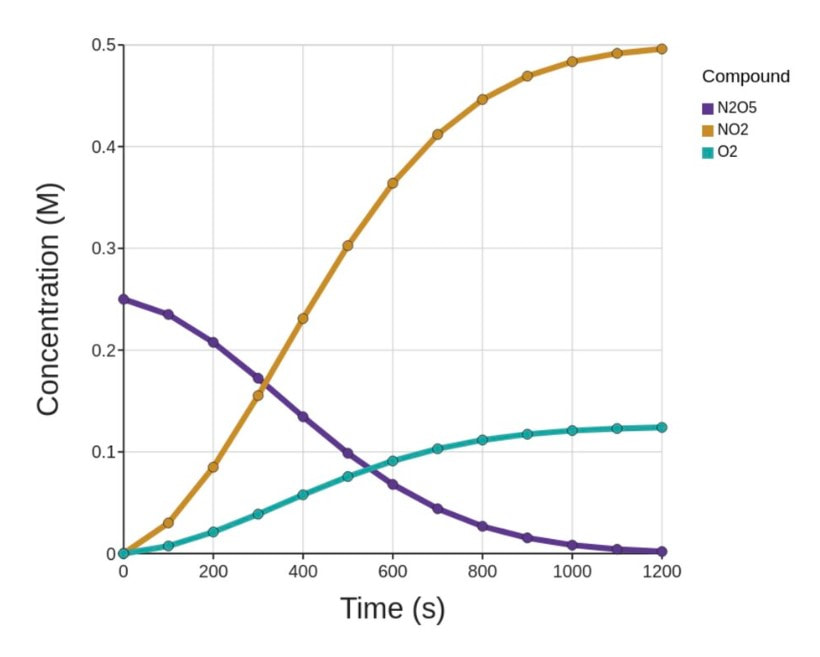
Two things to notice here:
- Dinitrogen pentaoxide is decreasing. Nitrogen dioxide and oxygen are both increasing.
- The rate of increase of oxygen is half that of nitrogen dioxide because the mole ratio is 1:2. The rate of increase of nitrogen dioxide is half the decrease of dinitrogen pentaoxide because the mole ratio is 2:4. The ratio of change varies based on the molar ratio of the balanced chemical reaction.