Solids
|
Structure of Solids
Solids exist because the intermolecular forces or bonds that hold it together are so strong (compared to the kinetic energy) that the molecules have no ability to actually move. They can vibrate, but they cannot translationally move. When the molecules or atoms are locked into a position, the structure can be crystalline or amorphous. Crystalline means that there is a repetitive pattern to the structure. Most ionic compounds will have a specific shape structure that will repeat over and over again. Examples of some of the structures that are possible are to the right. It is not necessary to know the specific names and their shapes. It is more important to know that crystals will repeat a predictable pattern over and over again. |
|
Amorphous solids are solids that have no predictable pattern to the way the molecules are held to one another in the solid. For instance, it is said (and is for the most part true) that no two snow flakes are alike. When water freezes, the "crystals" that are formed are not consistent. Due to the hydrogen bonding's strong attraction that can attract to many molecules, it is somewhat random the way water crystals form. You can see patterns, but different conditions can change them. For this reason, water is considered amorphous.
Because intermolecular forces vary in their strength, most molecular compounds are amorphous. They may very well form a crystal, but it is inconsistent. Or it may not form a discernible pattern. Either way, if there is no pattern or an inconsistent one, the solid is considered amorphous. |
Types of Solids and the Forces that Hold Them Together
Molecular Compounds & Nonmetals: As we talked about before, intermolecular forces are the primary forces that hold molecular compounds together as a liquid and a solid. When the temperature gets cold enough, the kinetic energy of the particles become so low that the intermolecular forces (hydrogen bonding, dipole-dipole and London dispersion) can lock the molecules into place to form the solid. These are the weakest of all the forces in this list, and as a result these materials will (in general) have the lowest boiling points.
Molecular Compounds & Nonmetals: As we talked about before, intermolecular forces are the primary forces that hold molecular compounds together as a liquid and a solid. When the temperature gets cold enough, the kinetic energy of the particles become so low that the intermolecular forces (hydrogen bonding, dipole-dipole and London dispersion) can lock the molecules into place to form the solid. These are the weakest of all the forces in this list, and as a result these materials will (in general) have the lowest boiling points.
|
Ionic Compounds: Up until now, we have discussed ionic compounds as if they are a discreet (individual) molecule. For instance, sodium chloride is NaCl and I have led you to believe that an NaCl molecule can be independent of all others. This is a little bit of a falsehood.
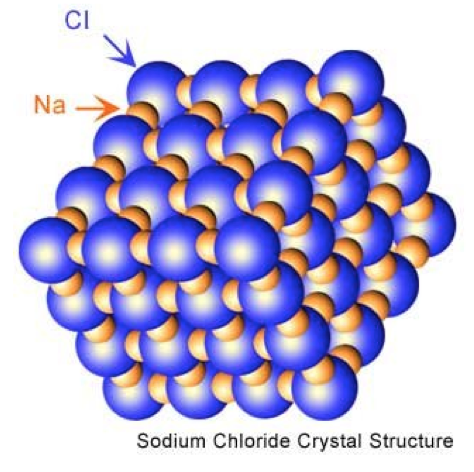
All ionic compounds are solid at room temperature because the attraction from a positive ion to a negative ion is so strong that it is difficult to break it. In fact, the attraction from a positive sodium ion is so strong, it attracts all the negative chlorine ions around it. So, if we look at one sodium ion, it is not bonded to anyone chlorine ion. It's bonded to all of them. Which means the crystal it forms is extremely strong, and changing sodium chloride into a liquid where the discreet molecules can flow pass one another requires a very high temperature. In the picture to the left, notice that every sodium is touching a chloride. A single sodium is bonded to as many as 6 chlorines, making the sodium just as attracted to any of the chlorines. This holds the crystal together with a strong force, that makes it very hard to melt or boil. High temperatures will be necessary to melt or boil an ionic compound. |
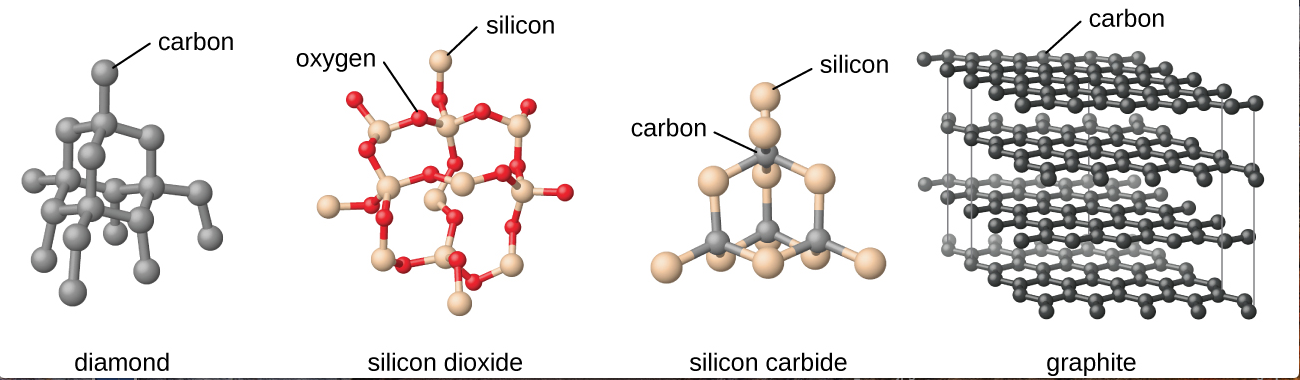
Covalent Network Solids: This type of solid primarily exists in elemental carbon (graphite, diamonds), silicon, and silicon dioxide (quartz). It can also occur in some compounds of boron, and in elemental germanium and boron. In these materials, the crystals are formed by covalent bonds between the materials. For instance, in a diamond, each carbon is bonded to 4 other carbons, and each of those carbons are bonded to 4 other carbons and so forth. All those covalent bonds hold the solid together very strongly, which results in a very high boiling point - even higher than ionic compounds. Some examples of the crystals of covalent network solid are below:
|
Metals: Metals, in their solid or liquid forms, do not have any of the above listed forces between them. They could have some London dispersion forces, but that is disputed, considering their boiling points are so high. So, another theory has been established for metals. The theory is called metallic bonding.
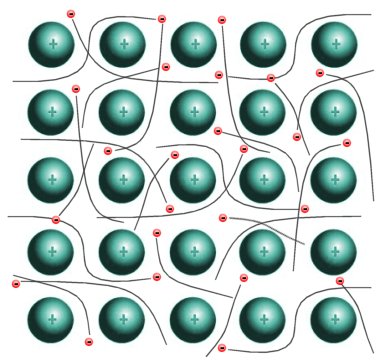
Metals are good at conducting electricity. This theory for metallic bonding takes that property and runs with it. Metals are held together by a "sea of delocalized electrons", which means that when metal atoms get together, they transfer their electrons amongst themselves. At any one time, the metal atom has however many electrons it needs to balance the protons, but the electrons are constantly in motion amongst the nuclei. This mutual need and transfer of electrons is what holds the metal atoms together, or metallic bonding. The electrons are in constant motion around the grid of atoms, and since there is a constant attraction to all the electrons, the metal stays together. Metal boiling points are pretty high, but the melting point varies widely. |