Applications of Intermolecular Forces
Now that we have established that intermolecular forces are the forces that keep molecules together in solids and liquids, we can discuss some of the effects of these forces. Depending on the strength of these forces within a liquid, that can affect several properties of the substance.
|
Viscosity
Viscosity is the resistance to flow of a liquid. Essentially, when a substance flows from one place to another slowly, this substance is considered to have a high viscosity. There is no direct measurement for viscosity, but we can compare viscosities of different liquids doing an experiment like the one to the right: When a liquid is poured, it will have a rate of flow. In the picture to the right, the honey has the highest viscosity. It moves to the bottom of the screen very slowly. The dish soap has the lowest viscosity. It moves down the screen very quickly. |
Viscosity is dependent on the intermolecular forces. The stronger the forces are, the slower the particles of the liquid can slide past one another. In these high viscosity liquids, the particles are so tightly held together that the liquid stays clumped and moves slowly.
Temperature does have an effect on viscosity. When you pour syrup out of a bottle, if the syrup is cold, the flow will be slow - high viscosity. If you heat up the syrup, it will flow faster. This is because the warmer syrup molecules have more energy due to the heating. The additional energy allows the molecules to move more freely. The intermolecular forces are the same strength. The molecules are moving faster, so the IF have less effect and can slide past one another more quickly and cause a faster flow.
Temperature does have an effect on viscosity. When you pour syrup out of a bottle, if the syrup is cold, the flow will be slow - high viscosity. If you heat up the syrup, it will flow faster. This is because the warmer syrup molecules have more energy due to the heating. The additional energy allows the molecules to move more freely. The intermolecular forces are the same strength. The molecules are moving faster, so the IF have less effect and can slide past one another more quickly and cause a faster flow.
|
Surface Tension
Another application of intermolecular forces is surface tension. Surface tension is why some bugs can walk across water without sinking into the water, and why water "beads" up on a penny or a leaf. You can see the surface tension in the picture to the right: If you look carefully at the bug and the paper clip sitting on the water, you will notice that the water looks bent. That's because it is. In these instances, the bug and paper clip are not floating, they are sitting on the surface of the water. When something floats, some of the material is actually below the surface of the water. In these cases, they are actually sitting on top of the water, like a pencil on a desk. |
|
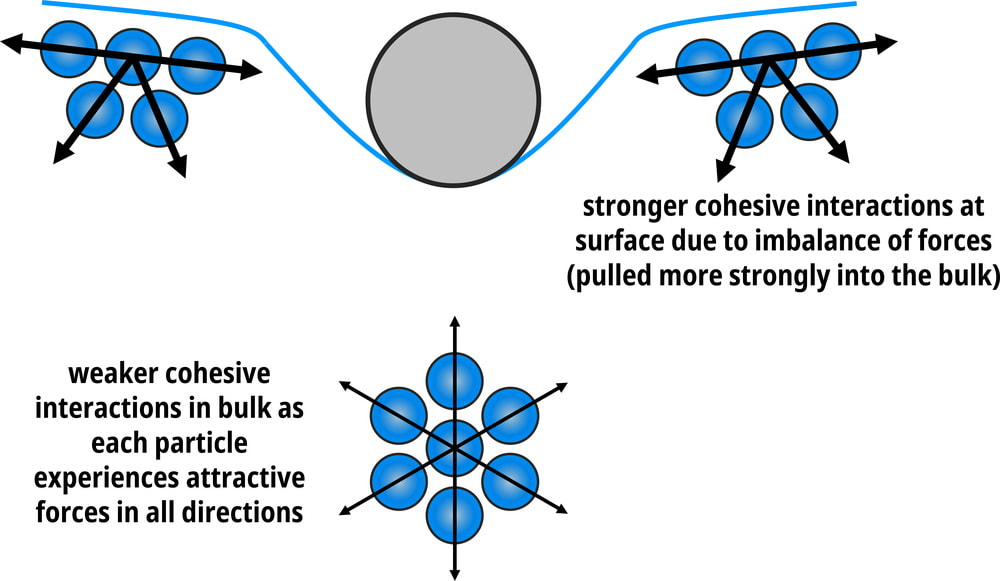
How does this happen? Surface tension is actually due to intermolecular forces being unbalanced at the surface of the liquid. Down in the liquid, each liquid molecule is being pulled equally with intermolecular forces in all directions by other liquid molecules completely surrounding it.
At the surface, the forces are unbalanced. There is a tension amongst the liquid at the top that pulls down into the liquid more, rather than up. This causes a "film", if you will, at the top that requires a little more force to break. That means a lighter object with more surface area may not break through the unbalanced forces and sit on top of the surface. If you break through the forces, though, since below in the liquid this tension doesn't exist, the object will sink all the way to the bottom. As with the viscosity, as the particles move faster, surface tension will decrease. So a higher temperature will decrease surface tension. |
|
Vapor Pressure
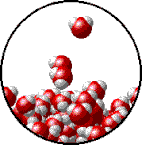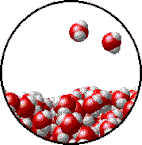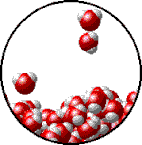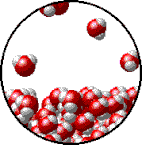
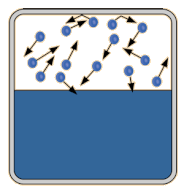
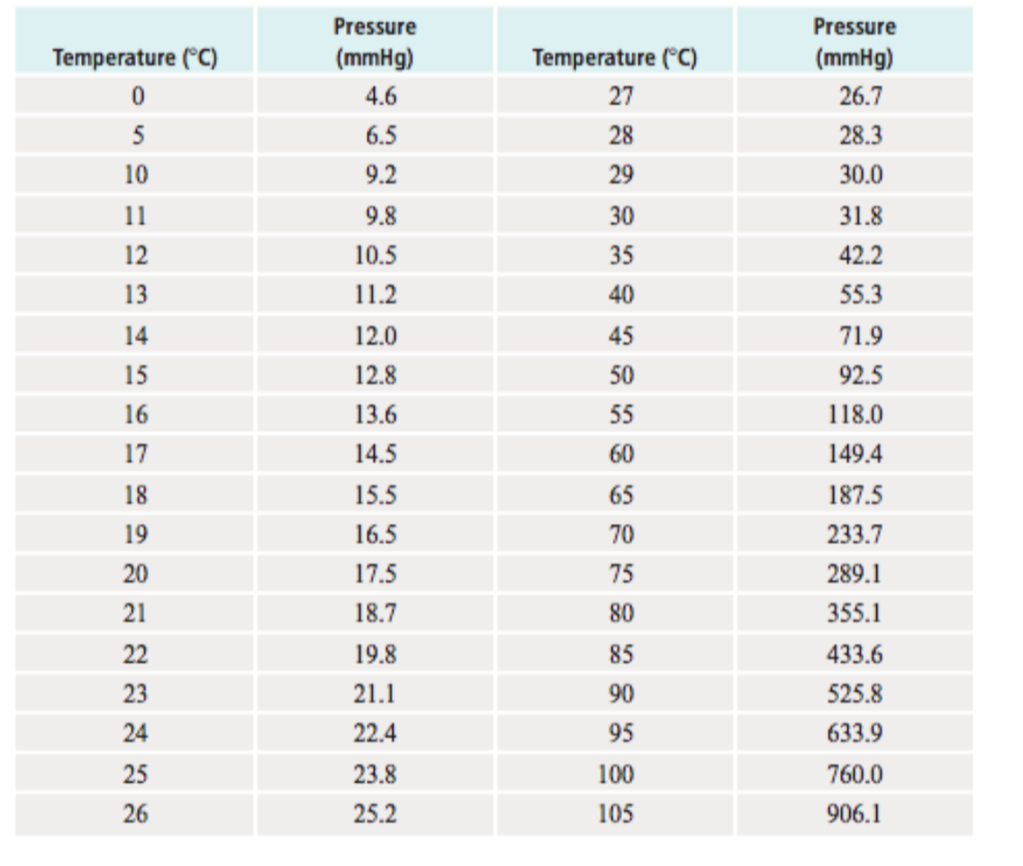
Vapor pressure is the pressure exerted by a recently evaporated gas. When a liquid, say water, sits in an open container, some of the liquid is going to evaporate. Evaporation occurs because heat and light energy can give some (not all) of the liquid particles at the surface enough energy break intermolecular forces and escape into the gas phase. When that gas is now above the liquid, it has all the properties of a gas, including pressure. The partial pressure exerted by the gas is called its vapor pressure. Vapor pressure depends on two things: intermolecular forces and temperature. At the bottom of the page is a table that compares the vapor pressure of water at different temperatures. You will notice from the data that as temperature increases, so does vapor pressure. Which makes sense. If you make the particles of the liquid move faster by heating them up, more will be escape the liquid phase and become a gas. More gas particles mean a higher partial pressure of evaporated gas above the liquid. Boiling and Vapor Pressure
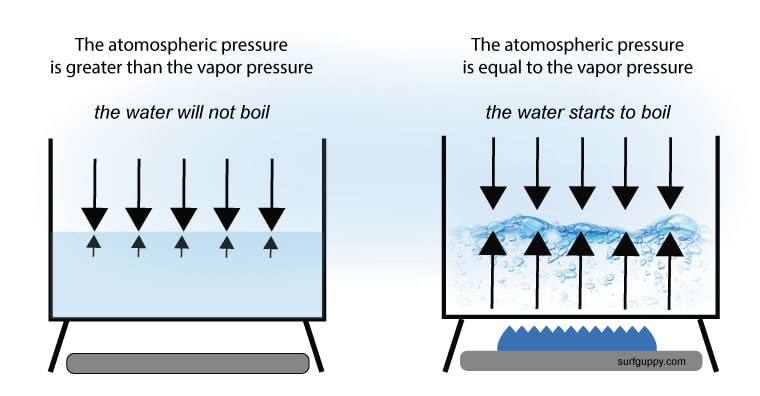
If you look at the data again, you will notice that for water, the vapor pressure of water reaches 760 mmHg at 100 OC. Now, we also know that water boils at 100 OC. That is not a coincidence. It's actually a causality. Boiling temperature is actually defined as the temperature at which the vapor pressure is equal to 1 atmosphere (760 mmHg) of pressure. A substance will boil when the vapor pressure leaving a liquid is equal to the atmospheric pressure pushing down on it.
|
Also, that means that boiling temperature is not the same at every atmospheric pressure. For example, in Denver, where the atmospheric pressure is lower, the temperature needed to make vapor pressure equal to atmospheric pressure is lower. That means water will boil at a slightly lower temperature. In fact, you can make any liquid boil at just about any temperature, as long as you get the pressure pushing down equal to the vapor pressure trying to escape.