Intermolecular Forces
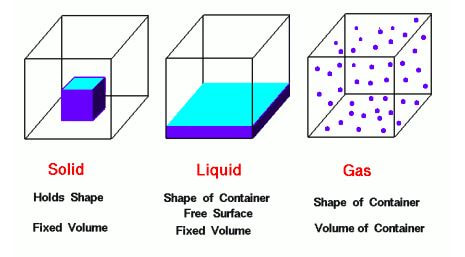
At room temperature, you can have different substances all at the same temperature, yet one may be a solid, another a gas and a third a liquid. Let's take a look at three substances: Gaseous oxygen, liquid water and solid lead, all at 298 K.
he gases molecules are independent of one another, moving around in random patterns and colliding based on the kinetic molecular theory. The solids and liquids, on the other hand, have molecules that are not independent. They are stuck to other version of the themselves. In water, the water molecules are all so close to on another that we can see the liquid water. In this instance, there must be something, some force, that holds the water molecules to each other. Otherwise, they would fly apart from one another like in a gas. So, what are these forces that hold the liquids and solids together?
Kinetic Energy and Attractive Forces
First, we need to look at the relationship between temperature and the state of matter. As we already know, as we cool a gas down, in most cases the gas turns into a liquid and then a solid. As the temperature decreases, the particles start to move slower. At some point, the particles end up moving so slow, they start to stick together in a liquid. The question becomes: Why do they stick together? They don't bond to one another. They just stick together as if they had some "glue" holding them together like two pieces of paper glued together. Then, when they slow down even more, the liquid becomes a solid, where they seem to be held together with even more "glue".
Kinetic Energy and Attractive Forces
First, we need to look at the relationship between temperature and the state of matter. As we already know, as we cool a gas down, in most cases the gas turns into a liquid and then a solid. As the temperature decreases, the particles start to move slower. At some point, the particles end up moving so slow, they start to stick together in a liquid. The question becomes: Why do they stick together? They don't bond to one another. They just stick together as if they had some "glue" holding them together like two pieces of paper glued together. Then, when they slow down even more, the liquid becomes a solid, where they seem to be held together with even more "glue".
|
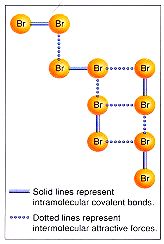
The "glue" is due to the attraction from one molecule to another. These attractions are caused by many factors, which we will look at below. But, regardless of the reason, the "glue" has a force which holds one molecule to another. The bonds within the molecules still exist, but now there is a force that holds one molecule to the next. We call these intermolecular forces. These are the forces that hold one molecule to the next, and when enough of these molecules are "stuck" together, a droplet of liquid will form. These intermolecular forces always have the same strength, and when the molecules slow enough (due to a decrease in temperature), they will have the strength to form "clumps", and when enough "clumps" "stick" together, then you will see liquid droplets form. The intermolecular force will hold together better as the molecules move slower. That's why solids are stronger than liquids, because the molecules are moving slower, so the forces can hold together better.
|
Types of Intermolecular Forces
Intermolecular forces are the forces that hold together covalent (molecular) compounds in order to form the liquid and solid states. There are essentially three kinds of these forces. The list below ranks them in order from weakest to strongest.
Intermolecular forces are the forces that hold together covalent (molecular) compounds in order to form the liquid and solid states. There are essentially three kinds of these forces. The list below ranks them in order from weakest to strongest.
|
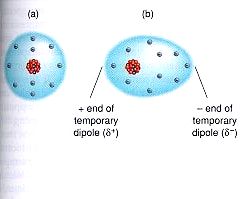
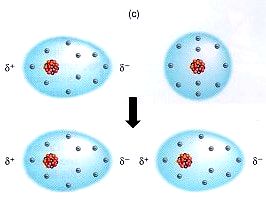
1) London Dispersion Forces: These are the forces that hold together nonpolar molecules. Nonpolar molecules are not dipoles, meaning they do not have partially charged ends. We can think of them as neutral (a). However, at extremely low temperatures, the molecules move very slowly. In fact, they move so slowly, the electrons begin to slow down. When they slow down, the electrons don't spread out evenly and start accumulating on one side of the nucleus. This creates something called a temporary dipole, where one side of the molecule (the one with more electrons) is negative and the other side (with fewer electrons) is positive (b). This temporary dipole can then create other temporary dipoles (sometimes called induced dipoles) which can then attract one another (c). This attraction can be repeated over and over which can clump enough molecules together to form a solid or liquid.
Since these are weak forces, the temperature has to be really low for them to condense or freeze the material. Therefore, nonpolar substances have low boiling points. In general, larger molecules have greater dispersion forces. The higher the molar mass or the more stretched out the molecule, the greater the dispersion forces. For instance, hydrogen (H2), will have very weak forces and a very low boiling point. Carbon tetrachloride (CCl4), has a much larger molar mass and will have a considerably higher boiling point.
|
|
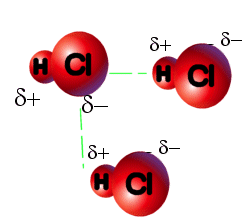
2) Dipole-Dipole Forces: These forces hold together polar covalent molecules. These forces are very similar to dispersion forces, except the molecules already start as dipoles. A polar covalent molecule is another word for dipole. These molecules already have a positive and negative end to the molecule. When two dipoles come close to one another, the opposing polar ends will attract one another. As they attract, they "clump", and create solids or liquids.
Molecules with dipole-dipole forces will have higher boiling points (in general) than molecules that have only dispersion forces. Also, any molecule with dipole-dipole forces will also contain London dispersion forces. The forces are additive from here on. In the image to the left, the H is partially positive and the Cl is partially negative. The H of one HCl molecule is attracted to the Cl of another HCl molecule. The green dotted line shows the attraction between the two molecules. |
|
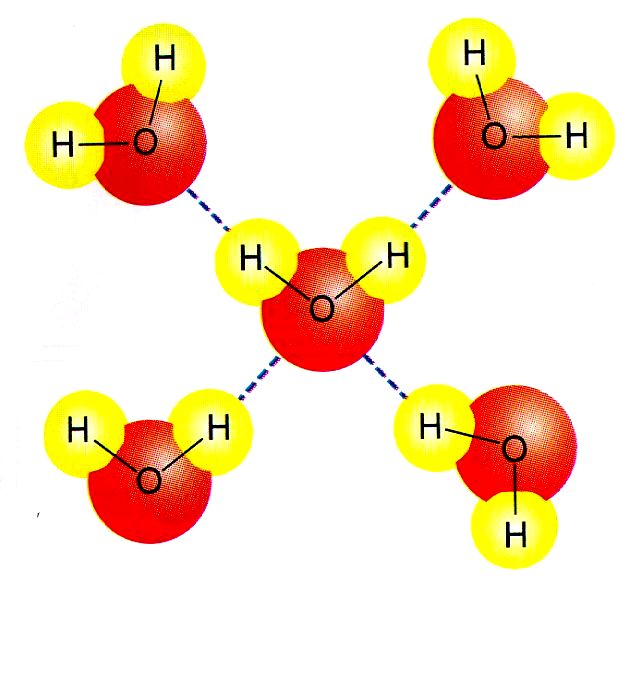
3) Hydrogen bonding: Not really bonding, but just called that. These forces exist in molecules that have a H-F bond, a H-O bond or a H-N bond. These bonds are very polar, which create very polar molecules. These intermolecular forces are dipole-dipole forces, but are so strong they are given their own special name. When these forces exist in a molecule, then dipole-dipole and dispersion also exist. These substances would have very high boiling points in comparison to dispersion and dipole-dipole.
|